Preclinical models are crucial to the drug discovery process because they help scientists find promising compounds, or “hits,” using high throughput drug screening assays. To find the preferred biological activity, the process screens various compounds against human and/or nonhuman model systems.
After in vitro hit identification, nonhuman animal models are used to assess toxicology and efficacy within the complexity of an entire organism due to ethical and financial considerations. Mice or zebrafish (Danio rerio) can be used to establish a desirable effectiveness profile as early as possible, despite their flaws.
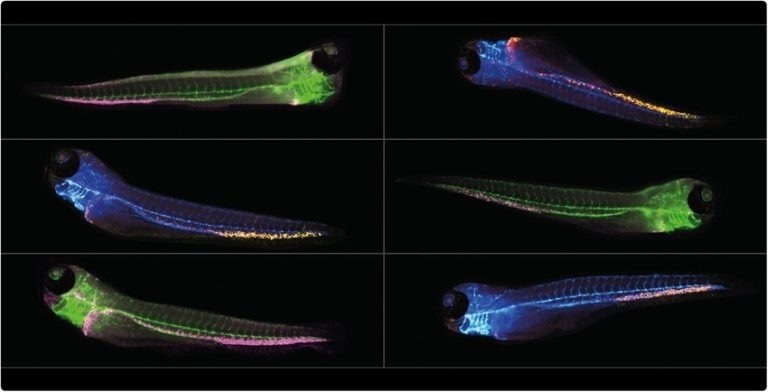
Image Credit: IDEA Bio-Medical Ltd.
The Importance of In Vivo Drug Testing
Preclinical drug screening is often carried out using in silico, in vitro, and in vivo systems. The first two techniques can offer crucial information for locating novel compounds that cause desired reactions. For example, monolayer cultures are very good at identifying tissue-specific toxicity phenotypes.
The study of how neighboring cells can influence the response to pharmaceuticals is also made possible by new 3D cell culture techniques. They are unable to offer the level of detail required to comprehend more advanced mechanisms resulting from interactions between various tissues. In vivo animal models are needed for that.
The recognized animal model for biomedical studies and clinical trials is the mouse. Their physiological resemblance to humans is just one of the many factors that make them particularly suitable for laboratory settings. However, the zebrafish embryo has overturned the dominance of using mammalian animal models to investigate the causes of human disease and conduct drug testing.
Why Use Zebrafish Models Instead of Mice?
A potent technique for examining small molecule interactions in living multiorgan systems is chemical screening in zebrafish. Compound toxicities, metabolic changes, pharmacokinetics, and cell niche modulations are examples of observables. Although mice and humans share a closer evolutionary relationship, studies have found that roughly 70% of human genes share at least one zebrafish orthologue.
Zebrafish are an outstanding platform for cancer and therapeutic research because they also have a wealth of advantageous traits. They can be housed in shoals of hundreds and can be kept in smaller spaces than mice, saving space and cost. The zebrafish genome can be modified genetically using CRISPR to add cell-specific readouts like fluorescence.
Additionally, they produce hundreds of eggs at a time during their reproductive cycle, which is measured in days as opposed to weeks. As a result of these benefits, libraries of specialized zebrafish lines are constantly expanding. Zebrafish are also easier and less expensive to maintain than mice.
Zebrafish screening has advantages that go beyond purely practical ones, however. Due to their optical transparency, zebrafish embryos make a remarkable testing medium for drugs. A variety of imaging modalities can be used to directly visualize fluorescently labeled tissues in transgenic zebrafish embryos and, using transmitted light for label-free imaging studies, many organs can be directly visualized.
The size of the embryos amplifies the benefits for microscopy as they can fit within multi-well plates to potentially increase experimental throughput.
Challenges of zebrafish screening
Zebrafish have many advantages for drug screening assays, but high throughput screening presents significant difficulties. It can be challenging to automate the data acquisition and analysis workflows when using microscopy as a tool. The zebrafish embryo must be positioned correctly to produce insightful images.
If not, the anatomy of interest might not be completely visible or within the visualized focal plane. Presently, it is a regular practice for scientists to manually arrange embryos one at a time into preferred lateral or dorsoventral orientations before imaging.
Automating image analysis encounters a significant bottleneck when imaging a large number of zebrafish. The size of the challenge is exacerbated by the use of multi-well plates. The fact that these fish are living, three-dimensional objects present the first challenge.
It is, therefore, unlikely that they will maintain a perfect positioning within the wells while under anesthesia because they can roll and drift. It is impractical to manually scan through the images on a large-scale automated screen to weed out the fish that are positioned incorrectly.
The ability to focus on one particular anatomical region of the zebrafish embryo while ignoring others presents a crucial image analysis challenge. For instance, standard fluorescence analysis using thresholding detects bright spots elsewhere in the fish when examining cells in the tail, either from cells outside the anatomy of interest or from autofluorescence.
Currently, it is a common practice to either manually select the region of interest or ignore the visible anatomy and simply assess fluorescence images using intensity thresholding. While the latter prevents automated workflows and limits the size of drug libraries that can be studied, the former decreases the information extracted from each embryo.
Solutions For Zebrafish Screening
For in vivo zebrafish screening, IDEA Bio-Medical provides a cutting-edge deep learning-based image analysis solution. It is a cost-effective, simple-to-use system created especially for quick zebrafish image-based screening.
Zebrafish imaging on the WiScan Hermes
Zebrafish imaging on the WiScan Hermes. Video Credit: IDEA Bio-Medical Ltd.
About IDEA Bio-Medical Ltd.
IDEA Bio-Medical is founded in 2007 through a partnership between YEDA (the Weizmann Institute’s commercialization arm) and IDEA Machine Development (an innovation hub).
We specialize in automated imaging systems and image analysis software, offering a broad range of biological applications based on the company’s unique algorithms library. The company is developing novel image-based screening platforms for the pharmaceutical industry and medical centers, dedicated to broadening the scope of personalized medicine.
Our WiScan Hermes system incorporates the most advanced technologies currently available in the machine vision field, integrated with engineering methodologies of high reliability and quality at the level of semi-conductors and digital printing industries, which are the specialty of our mother company, IDEA Machine Development Design and Production Ltd.
Sponsored Content Policy: AZO Life Sciences publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of AZO Life Sciences which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.