Glioblastoma is a very aggressive form of cancer that affects 3 in every 100,000 individuals, encompassing the bulk of malignant tumors. Like all astrocytomas, their tumor formation is dotted along the brain and spinal cord.
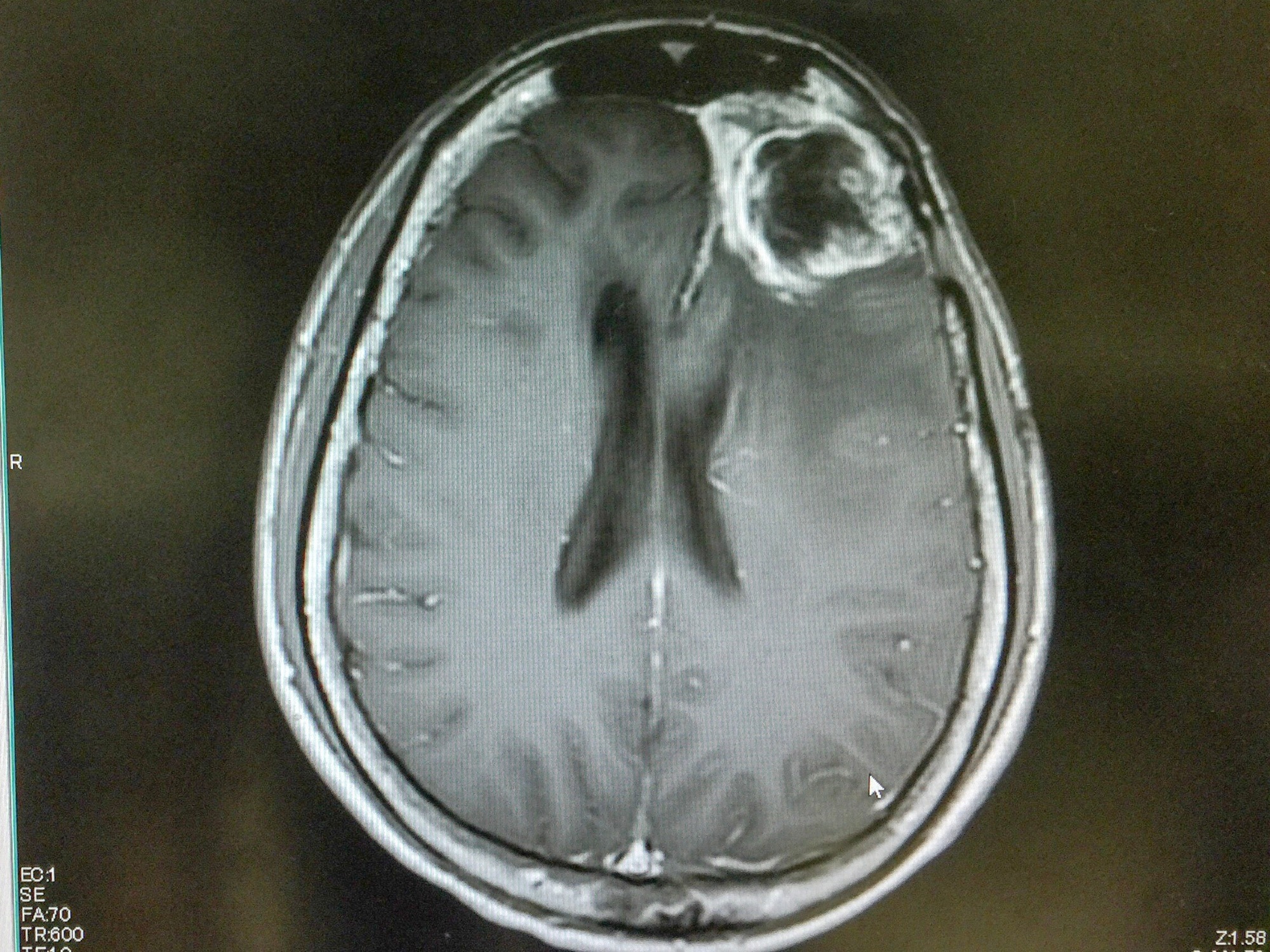
Image Credit: O_Akira/Shutterstock.com
However, all glioblastomas are stage IV tumors because of their high malignancy and rapid growth. These tumors often originate in one of the two cerebral hemispheres, invading nearby tissue until it crosses the corpus callosum. Though magnetic resonance imaging (MRI) is the gold standard for tumor diagnoses, a series of spectroscopy tests may be necessary or even preferred when assessing the chemical metabolism of the tumor or tumors.
Initial Diagnosis of Glioblastoma Tumors
A CT scan is obtained if one presents symptoms such as headaches, seizures, severe confusion, and sudden loss of vision. An MRI often follows this. Though this methodology is the most practiced, MRIs may not always be specific when undergoing tumor analysis. To correctly identify a glioblastoma from other forms of gliomas, or various other masses, a severe contrast enhancement of the tumor must be shown. This explores a region of focus, which may result in a noisy signal and a loss of understanding of the metabolite consumption/ presence within these masses.
This is where localized proton spectroscopy would come into play. Though spectroscopy can be performed using phosphorus (P31), or even sodium (Na11), the most widely used form would be proton (H1) MR spectroscopy, which fits with the highest sensitivity and best ease of implementation.
Single Voxel (SV) techniques incorporating the Point resolved spectroscopy (PRESS) and the Stimulated echo acquisition mode (STEAM) Spectroscopy
Magnetic resonance spectroscopic imaging (MRSI) can be employed using varying settings, all suited for one approach or another. The classification of tumor masses among patients is associated with either single-voxel or multi-voxel techniques of MR spectroscopy.
The Single-voxel method will sequentially record a spectrum from different brain regions, while multi-voxel techniques will record spectra from multiple regions at once, mapping out the distribution of metabolites at a single point in time. The former is the simplest and easier to interpret, while the latter can quantify chemical biomarkers that may interest the physician. Single voxel techniques may incorporate a PRESS method or the STEAM method.
PRESS Method: Though assessment and diagnosis via surface NMR detection are considered inadequate for deep-lying, proximal organs, selecting planar volumes is apropos when using this single excitation pulse. Point-resolved spectroscopy (PRESS) is a method that improves spatial selectivity by producing a spin echo. The surface coil used to localize masses does have an extremely high sensitivity, which can result in a noisy NMR signal, though this effect can be somewhat alleviated by adjusting the flip angle beyond P/2. This modus is employed when surveying samples with long spin-spin relaxation times.
STEAM method: Stimulated echo acquisition mode (STEAM) NMR fits the requirements of H1 NMR almost perfectly when conducted in vivo. This specified single voxel method employs three slice-selective 90o RF pulses, which minimizes power consumption and allows for more accurate determinations when taking short T1-weighted or longer T2-weighted images. This method is employed when assessing shorter spin-spin relaxation times.
Multi-Voxel Techniques in MRSI Imaging
Spectroscopy via MRSI is useful when assessing glioblastoma tumors for very select reasons. While most astrocytomas are dense, have elevated levels of choline, and have a relatively high membrane turnover, glioblastomas often differ. Many studies have provided evidence of grade IV glioblastoma expressing lower levels of choline and lower densities because of their necrotic cores (associated with diminished levels of all metabolites).
The heterogeneity of these tumors makes traditional MRI scans less accurate, as they resort to a region of interest. MRSI fitted with multi-voxel techniques is better in this case because it can evaluate metabolic heterogeneity in both high-grade and low-grade gliomas.
By using MRSI in this way, we can differentiate between spectrum from necrotic or non-necrotic cores, assess tumor invasion amongst surrounding grey matter, and offer versatile data regarding tumor metabolism. A study by Hourani R et al. determines that the most discriminant feature between brain lesions and brain tumors was the ratio of choline to creatine (Cho/Cr); also an apt measurement when assessing high and low-grade tumors.
By fitting this method to state-of-the-art 3T MRIs, high- and low-grade gliomas may be assessed. Different forms of neoplasia can be assessed this way, depending on how data collection and analysis techniques are performed on the part of the physician.
This does not mean that this technique is foolproof and still carries some inherent risk. Brain lesions are inconsistent in nature and may overlap with other tumor types. There is also variability in analytical technique and interpretation depending on the oncologist.
For these reasons, MRSI alone may not come with as high confidence as some may think when used in a clinical setting. However, because of the former clauses stated above, it is agreed upon by most health professionals that H1 MRSI can have promising results in differentiating brain lesions from glioblastomas.
Sources:
- Horská, A., & Barker, P. B. (2010). Imaging of brain tumors: MR spectroscopy and metabolic imaging. Neuroimaging clinics of North America, 20(3), 293–310. https://doi.org/10.1016/j.nic.2010.04.003
- Posse S, Otazo R, Dager SR, Alger J. MR spectroscopic imaging: principles and recent advances. J Magn Reson Imaging. 2013 Jun;37(6):1301-25. doi: 10.1002/jmri.23945. Epub 2012 Nov 27. PMID: 23188775.
- Hourani R, Horská A, Albayram S, Brant LJ, Melhem E, Cohen KJ, Burger PC, Weingart JD, Carson B, Wharam MD, Barker PB. Proton magnetic resonance spectroscopic imaging to differentiate between nonneoplastic lesions and brain tumors in children. J Magn Reson Imaging. 2006 Feb;23(2):99-107. doi: 10.1002/jmri.20480. PMID: 16374884.
- Jens Frahm, Klaus-Dietmar Merboldt, Wolfgang Hänicke, (1969) Localized Proton Spectroscopy Using Stimulated Echoes, Journal of Magnetic Resonance; 72-3 502-508 ISSN: 0022-2364 https://doi.org/10.1016/0022-2364(87)90154-5.
- Albulescu, R., Petrescu, A. J. , Sarbu, M., Grigore, A., Ica, R., Munteanu, C. V. A. , Albulescu, A., Militaru, I. V. , Zamfir, A., Petrescu, S., & Tanase, C. (2019). Mass Spectrometry for Cancer Biomarkers. In (Ed.), Proteomics Technologies and Applications. IntechOpen. https://doi.org/10.5772/intechopen.85609
Last Updated: Nov 11, 2022