While the genetic code is known to impart information on cellular processes, the associations between regions of DNA and their phenotype are still being made. The genetics of human health and disease is often more complicated than a causative single gene; a complex interplay between genetics, epigenetic modifications, and mitochondrial DNA underpins varied expression of genes that can lead to a diseased state.

Image Credit: WinWin artlab/Shutterstock.com
The online mendelian inheritance in man (OMIM) celebrates 50 years of cataloging human genes and genetic disorders. As of October 29th, 2021, 4,533 genes with phenotype-causing mutations have been identified. Though disease pathogenesis is often more complex than simple monogenic etiology, single-gene disorders and traits account for ~3/4 of OMIM registered phenotypes. Of a total of 7,014 phenotypes, 5,944 single disorders and traits are registered in OMIM.
As genomics and genetics advance, linking genetic components and disease states may become easier. By comparing to the mapped “healthy” sequence, inherited or acquired variations in the genetic code can be more easily associated with a disease phenotype. Unsurprisingly, it is often more complicated than it sounds. Physical, nutritional, and emotional environmental stressors can impart complex patterns on DNA that alter the expression of a gene. These patterns are termed epigenetic markers.
Epigenetic markers modify the structure of DNA
The epigenome refers to a combination of processes that regulate the expression of genes, without altering the genetic code. Through histone modifications, nucleosome positioning, and DNA methylation, epigenetic “markers” can impart instructional information that can alter the expression of genes.
These markers, or chemical compounds, are added to DNA bases and histone proteins to alter the spatial conformation of chromatin. As a result, proteins interactions with the DNA are inhibited or promoted; altering the transcription of genes in the region.
DNA methylation signals for gene inactivation
In eukaryotes, regions of cytosine and guanine dense DNA can be found upstream of many genes, termed CpG islands. By targeting copies of the sequence 5’-CG-3’ in CpG islands, DNA methyltransferase converts cytosine in the regions to 5-methyl-cytosine.
Methylation then attracts components of the histone deacetylase complex to modify surrounding chromatin. This process can occur de novo or during DNA synthesis, ensuring the daughter DNA strands retain the correct methylations patterns.
Chemical modification of histone proteins
Methyl-CpG-binding protein components of the histone deacetylase complexes (HDAC), bind to 5-methyl-cytosine in CpG islands to repress gene activity. By removing the acetyl group from histone tails, HDAC increases the histone protein’s affinity for DNA. This results in structural changes, such that the chromatin is packed more tightly; limiting the ability of transcription factors to bind DNA and thus silencing the adjacent genes.
Histone acetyltransferase (HAT), on the other hand, forms complexes to add acetyl groups. Acetylation of histone proteins is associated with a more open chromatin state, allowing for increased gene expression. The expression of genes can also be modified by post-translational methylation, ubiquitination, sumoylation, phosphorylation, biotinylation, and ADP-ribosylation of histone proteins.
The specific modification and the amino acid position on a particular histone protein will influence its function; with each specific modification governing the binding of specific effector proteins to influence cellular processes.
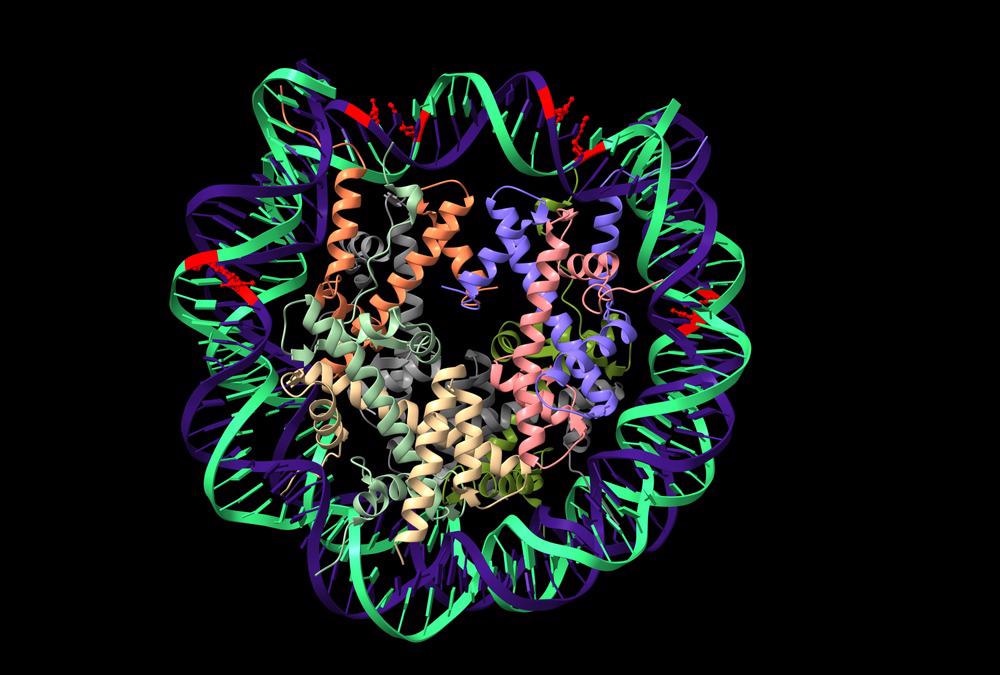
Image Credit: Volodymyr Dvornyk/Shutterstock.com
Nucleosome repositioning to allow for protein binding
The spatial arrangement of chromatin can also be altered by non-chemical modifications. Through three distinct methods of repositioning, the attachment site of DNA-binding proteins can be changed. Remodeling refers to changing the nucleosome structure while sliding and transfer refer to the physical movement of nucleosomes. Repositioning of nucleosomes, or altering the binding site of histone, is thought to support DNA damage response and repair.
By shifting along the same DNA molecule, sliding is able to displace the nucleosome; similarly, transfer results in the movement of the nucleosome to a non-abject segment, or a second molecule, of DNA. Remodeling complexes, such as SWI-SNF, detect the presence of histone modifications and modify nucleosomes in an ATP-dependent manner. Mutations and silencing in any of the components of the complexes involved in these processes can result in impaired or inappropriate DNA transcription and repair.
The influence of mitochondrial DNA on human health
Mitochondria contain their own set of maternally inherited DNA, which encodes thirteen peptides involved in oxidative phosphorylation and is critical to the production of cellular energy. Mutations and damage to mitochondrial DNA are associated with a variety of conditions, including cancers and neurodegenerative disease.
A recent analysis by Yuan et al. defined the mutational landscape of the small mitochondrial genome in cancers. This hopes to provide the foundation for translating mitochondrial biology into clinical applications. Of the 13 protein-coding genes, it was found that ND5 was the most frequently mutated in most cancer types; ND4 was most frequently mutated in prostate and lung cancers; COX1 was most frequently mutated in breast, cervical, and bladder cancers.
Sources:
- Tiffon, C. (2018) The Impact of Nutrition and Environmental Epigenetics on Human Health and Disease. Int J Mol Sci. doi:10.3390/ijms19113425.
- Brown, T.A. (2017) Genomes 4. Fourth edition. [Online] Yale J Biol Med. Available at: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5733849/. (Accessed on 1 November 2021).
- Sharma, P., & Sampath, H. (2019) Mitochondrial DNA Integrity: Role in Health and Disease. Cells. doi:10.3390/cells8020100.
- Reiko, W., et al. (2017). Nucleosome remodelling, DNA repair and transcriptional regulation build negative feedback loops in cancer and cellular ageing. Phil. Trans. R. Soc. doi: 10.1098/rstb.2016.0473.
- National Human Genome Research Institute. (2018). Genetic Disorders. [Online] National Institute of Health. Available at: https://www.genome.gov/For-Patients-and-Families/Genetic-Disorders (Accessed on 1 November 2021).
- OMIM. (2021). OMIM Gene Map Statistics. [Online] OMIM, John Hopkins University. Available at: https://www.omim.org/statistics/geneMap (Accessed on 1 November 2021).
Further Reading
Last Updated: Mar 4, 2022