Professor Sam Sheppard speaks to AZoLifeSciences about his research on intensive farming and how it could increase the risk of epidemics.
What provoked your research into the correlation between intensive farming and epidemics?
Having worked on infectious diseases for over a decade, I am aware that most bacterial pathogens exist principally as harmless commensal organisms (often in livestock).
Understanding what leads these organisms to become dangerous human pathogens is essential for safe sustainable food production and public health.
How has modern agriculture changed the distribution of animal species on Earth?
Livestock now accounts for 60% of all mammal biomass on earth, far surpassing the biomass of all wild mammals combined (4%).

Image Credit: Studio Peace/Shutterstock.com
Why has intensive farming increased globally over the years?
This has been important to meet the dietary needs and desire for meat and dairy products in an ever-growing global population.
Why is it important to look at the effects of intensive farming on public health?
Escalating livestock numbers and global trade have been linked with the emergence of zoonotic diseases, that transfer from animals to humans.
Understanding the emergence of these pathogens is necessary to guide modern farming practices and to minimize the risk for both animal and human health.
What is campylobacter bacterium and why did you choose to investigate this?
Campylobacter jejuni is the most abundant human bacterial pathogen found in cattle and many other animal species. It is also one of the most common global causes of food poisoning (gastroenteritis).
These organisms typically infect humans via consumption of contaminated meat and poultry, leading to widespread morbidity and, occasionally, death in vulnerable groups.
Campylobacter in the gut and manure of many millions of cattle presents a risk for spillover into the human food chain. However, little is known about how intensive farming may establish new transmission cycles among reservoir hosts and influence the evolution of livestock-adapted Campylobacter strains.
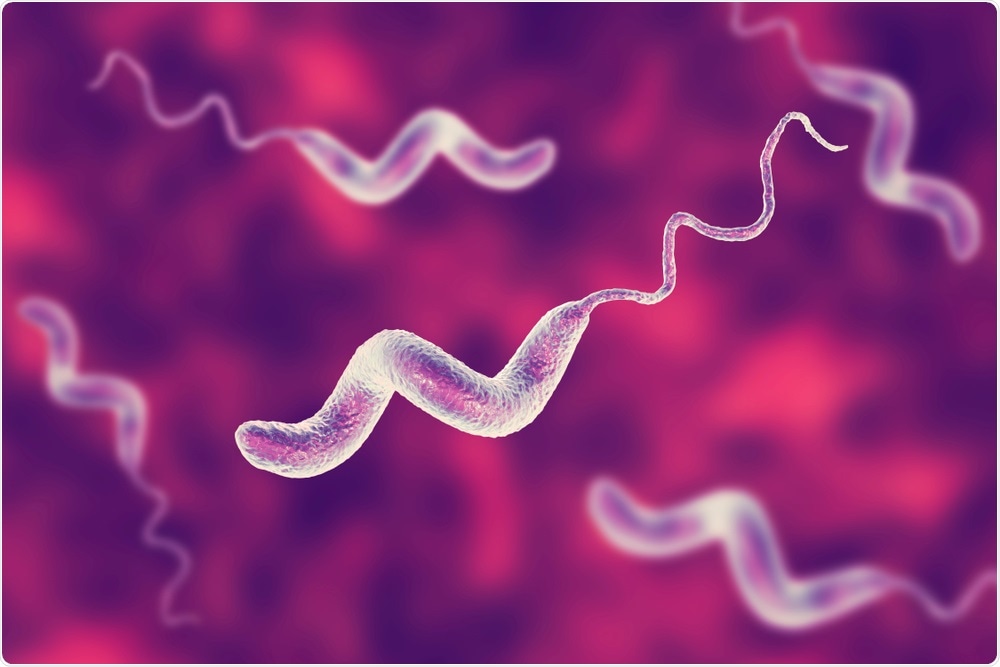
Image Credit: Kateryna Kon/Shutterstock.com
Why is campylobacter so resistant to antibiotics?
Antibiotics kill bacteria in many different ways. Antimicrobial resistance occurs when changes to bacterial genes confer new traits allowing the strains to survive.
Changes to the genetic code (genome) of bacteria occur in two ways: (i) by mutation introducing a change in the DNA sequence; (ii) by horizontal gene transfer, where the bacterium acquires genes from another strain and these genes recombine with the recipient strain genome – analogous to sex in animals.
Like some other bacteria, Campylobacter is highly recombinogenic. This means it can readily acquire antimicrobial resistance genes and will rapidly evolve resistance.
What could be done to reduce the transfer of this bacterium from cattle to humans?
We must gain a better understanding of the forces that promote the proliferation of Campylobacter and other zoonotic pathogens, and their transmission to humans.
This will require investment in research to identify the specific farming practices that promote pathogen transfer.
How could we increase the safety of farming methods to prevent the risks of outbreaks in the future?
This is a subject of much debate. Armed with the latest research findings livestock producers will be able to inform practices and target interventions.
What are the next steps in your research?
We are already developing our research beyond Campylobacter, to address complex questions in the ecology, epidemiology, and evolution of microbes and how this is related to the evolution of host/niche adaptation and virulence.
Where can readers find more information?
Lab website: https://sheppardlab.com/
PNAS cattle Campylobacter paper: https://www.pnas.org/content/early/2020/04/28/1917168117
About Professor Sam Sheppard
Sam is a former Wellcome Trust Fellow (Oxford University), Professor, and Director of Bioinformatics at the Milner Centre for Evolution (University of Bath).
He is also the Co-Principal Investigator of the original Medical Research Cloud Infrastructure for Microbial Bioinformatics (CLIMB, https://www.climb.ac.uk/) program, and holds honorary positions in Oxford and Swansea Universities and Public Health Wales.