Researchers from Germany’s Max Planck Institute for Plant Breeding Research (MPIPZ) and the University of Cologne have identified a new biochemical process that explains how immune proteins protect plants against invading microbes. Their results have been reported in Cell.
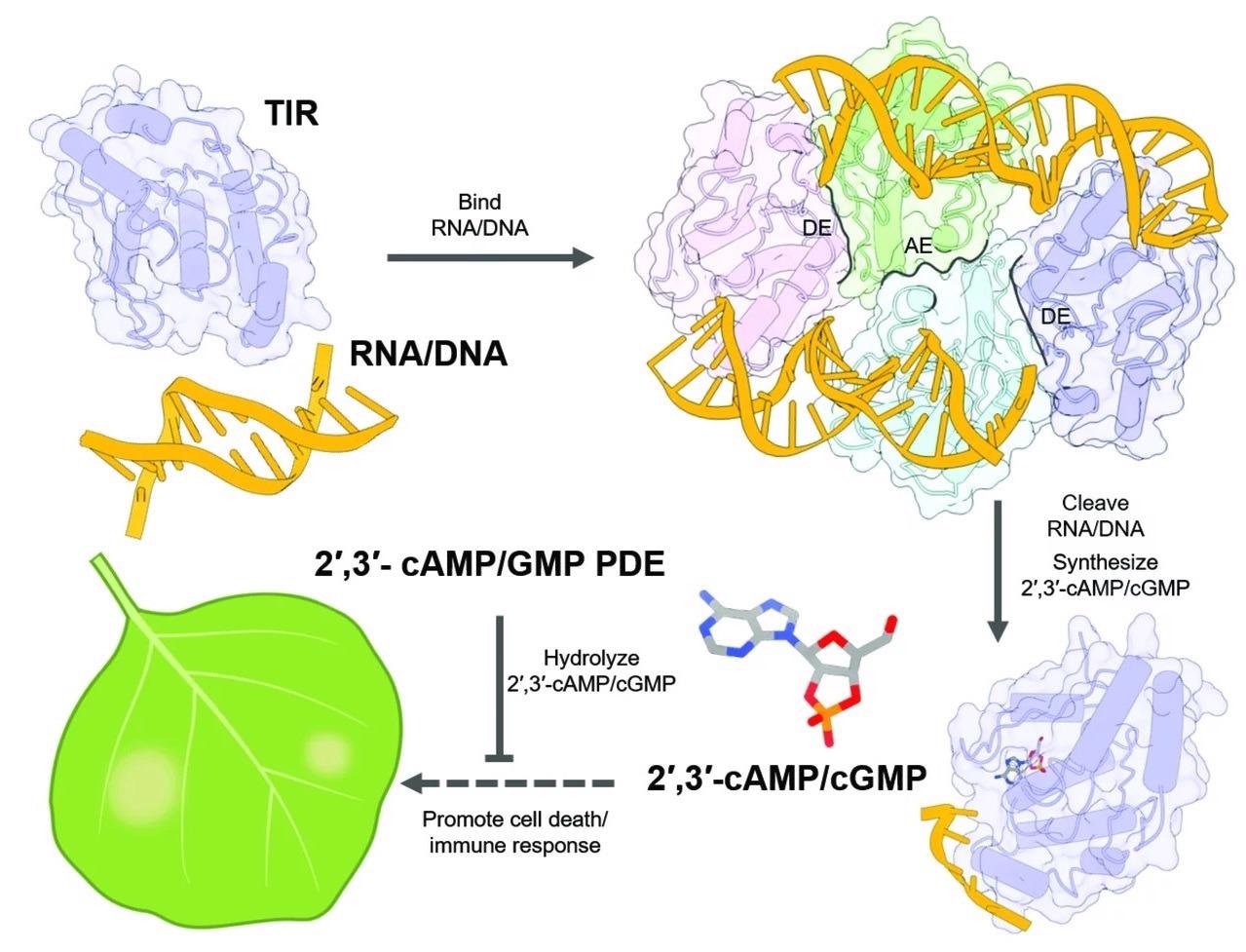
Image Credit: Max Planck Institute for Plant Breeding Research
Human immune systems are responsible for protecting from diseases caused by dangerous germs. Plants mount immune responses in the same way when they are invaded by dangerous microorganisms. Immune receptors, which indicate the presence of chemicals provided by alien pathogens and initiate defensive responses to resist the invaders, are key participants in these plant immune responses.
A subset of these immune receptors has specialized areas identified as toll-interleukin-1 receptor (TIR) domains and function as enzymes, special proteins that break down the molecule nicotinamide adenine dinucleotide (NAD+), an extremely abundant, multi-functional small molecule originating in all living cells.
Breakdown of NAD+ then activates more immune proteins, leading to the so-called “hypersensitive response,” a protective strategy that causes plant cells to die at locations of suspected infection as a means of protecting the plant overall.
However, investigations have revealed that, while NAD+ breakdown is necessary for plant defense, it is insufficient, implying that other mechanisms are required.
The researchers, led by the corresponding authors, Jijie Chai, of MPIPZ, the University of Cologne, and Tsinghua University in Beijing, China, Paul Schulze-Lefert from the MPIPZ, and Bin Wu from School of Biological Sciences, Nanyang Technological University, Singapore, examined the function of the TIR proteins and could show that these receptors not only broke down NAD+.
However, the TIR domains have an additional purpose: they process molecules with phosphodiester bonds, which are widely observed in RNA and DNA and are found in cells as big, linear single- or double-stranded molecules.
The authors used structural analysis to reveal that TIR proteins form separate multi-protein complexes for NAD+ or RNA/DNA degradation, demonstrating how one protein can play two roles. TIR proteins break RNA/DNA molecules by following the curvature of the strands and winding securely around them like pearls on a string.
TIR proteins’ ability to form two different molecular complexes is a trait shared by the whole immune receptor family. The activity of each enzyme is thus determined by the configuration of the TIR proteins.
The researchers proceeded to show that this function was insufficient for cell death, implying that specific tiny molecules produced by RNA and DNA breakdown were to blame. The researchers were able to determine the molecules as cyclic adenosine monophosphate/cyclic guanosine monophosphate (cAMP/cGMP), which are cyclic nucleotides found in all kingdoms of life.
Fascinatingly, rather than the well-characterized 3’,5’-cAMP/cGMP, the author’s investigation exhibited that the TIR domains were generating the production of the non-canonical 2’,3’-cAMP/cGMP, enigmatic “cousins,” whose accurate roles have so far been uncertain.
When they limited TIR-mediated production of 2? 3?-cAMP/cGMP, cell death activity was weakened, representing that the 2’, 3’-cAMP/cGMP molecules are significant for the plant immune reaction.
If 2’, 3’-cAMP/cGMP stimulate cell death in plants in reaction to infection, then it makes sense that their levels would be kept firmly in check. Certainly, the authors exposed that a known negative regulator of TIR function in plants, NUDT7, acts by reducing 2’, 3’-cAMP/cGMP.
Certain pathogenic bacteria emit similar negative regulators during infection within plant cells, and the researchers were able to show that these pathogen proteins decrease 2’, 3’-cAMP/cGMP as well.
This recommends that occupying microorganisms have grown clever strategies to disarm the 2’, 3’-cAMP/cGMP-dependent plant defense tool for their advantage.
Dongli Yu, one of the study’s three co-first authors, along with Wen Song and Eddie Yong Jun Tan, summarizes the implications of his research.
“We have identified a new role for the TIR domain of immune receptors in protecting plants against infection. Looking forward, identifying and characterizing the targets of 2’,3’-cAMP/cGMP will suggest novel strategies for making plants more resistant to harmful microbes and in this way contribute to food security,” Yu concluded.
Source:
Journal reference:
Yu, D., et al. (2022) TIR domains of plant immune receptors are 2′, 3′-cAMP/cGMP synthetases mediating cell death. Cell. doi.org/10.1016/j.cell.2022.04.032.