Neuronal cell death in the nervous system is a major contribution to neurodegenerative diseases. Despite occasional neuronal deaths during the process of aging, extensive neuronal cell death is rare in adults with a mature central nervous system (CNS).
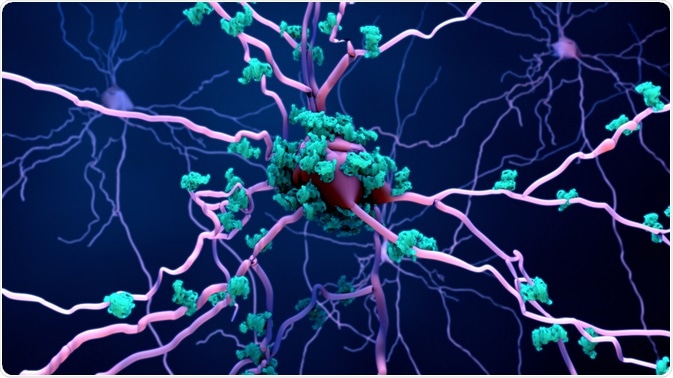
Image Credit: Design_Cells/Shutterstock.com
Nevertheless, there is an increased neuronal loss in patients with neurodegenerative diseases compared with healthy individuals of the same age group. The onset of neurodegenerative diseases often involves the formation of a pathological protein that aggregates and this places stress on neurons, potentially leading to neuronal death.
Understanding the pathological implications of neuronal cell death is essential for therapeutic development for neurodegenerative diseases.
Types of cell death
Apoptosis
Apoptosis is a form of programmed cell death where the cell shrinks without spillage of cellular content and DNA fragments.
It can be triggered extrinsically (e.g. detection of extracellular signals) or intrinsically (e.g. detection of internal stimuli such as DNA damage). The intrinsic pathway of apoptosis is also known as the mitochondrial pathway.
Cellular stress causes changes to the mitochondrial membrane leading to the loss of mitochondrial potential. This activates a group of proteins called caspases and leads to downstream morphological changes.
Necrosis
Necrosis is a form of cell death characterized by the swelling of cells and loss of membrane integrity. The leakage of cellular content usually leads to an inflammatory response.
Signals such as infection, radiation, reactive oxygen species, and Calcium ions overload can trigger necrosis. Ion pores are produced on the plasma membrane and the increased ion fluxes due to these new pores lead to osmotic imbalance and thus, bursting of the cell.
Role of cell death in the pathogenesis of neurodegenerative diseases
Alzheimer’s Disease (AD)
Characterized by memory loss, the most prominent symptoms of AD patients, AD disease progression is correlated to neuronal loss in the hippocampal region, which plays a major role in learning and memory.
There are conflicting reports of the mechanism of neuronal death in this disease, either by apoptosis or necrosis. Neuronal death can be caused by pathogenic protein aggregates of Tau and amyloid-beta.
Tau is a microtubule-associated protein and they are intrinsically unstructured. Pathogenic Tau proteins can interact with one another and form aggregate structures such as neurofibrillary tangles (NFTs).
Tau places stress on neurons by detaching from microtubules and damaging the cytoskeleton of neurons. Moreover, when detached Tau proteins aggregate in the cytosol, they are not degraded by the proteasome system and this leads to a systemic defect. Pathogenic Tau also induces re-entry of cell cycle and thus, leading mature neurons to cell death.
Amyloid-beta extracellular aggregate plaques are another hallmark of AD. However, the correlation between amyloid-beta aggregates and neuronal death is inconsistent across studies.
Nevertheless, it is proposed that neuronal exposure to amyloid-beta aggregates activates synapses, leading to an abnormally prolonged period of synaptic excitation, which is toxic to neurons and leads to cell death.
Image Credit: Atthapon Raksthaput/Shutterstock.com
Parkinson’s Disease (PD)
PD is characterized by neuronal cell death by apoptosis in the substantia nigra (SN) and severe loss of dopamine, a neurotransmitter with a role in pleasure and motor control. Symptoms include tremors and muscle stiffness.
α-Synuclein is the major pathogenic protein of PD. It is normally located at the tips of neurons and forms specialized structures called presynaptic terminals.
However, the pathogenic form of α-Synuclein aggregates, and it is unknown on a molecular level how α-Synuclein causes neuronal death.
Its expression in neurons has been linked to mitochondrial dysfunction and oxidative stress, and this implies a potential trigger for neuronal apoptotic death.
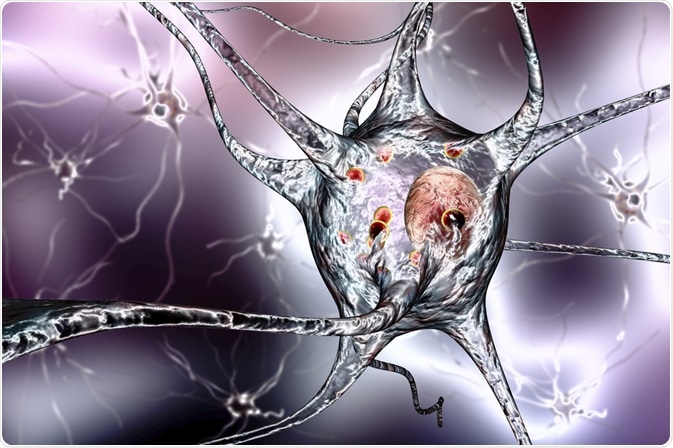
Image Credit: Kateryna Kon/Shutterstock.com
Huntington’s Disease (HD)
Huntington’s Disease is an inherited disease that causes uncontrolled movements. The loss of neurons in areas of the brain leads to the upregulation of excitatory signals between neurons in the motor circulatory and thus uncoordinated muscle movements.
Huntingtin protein, the cause of HD, contains trinucleotide CAG repeats in its gene. This repeat is unstable and could be duplicated and expanded during DNA replication. This leads to a loss of function of the protein and transforms the protein into its pathogenic form.
The mutant huntingtin protein aggregates intracellularly and traps other cellular proteins, such as transcriptional factors, plasma membrane, and cytoskeletal proteins, to form clumps. This negatively impacts neuronal functions.
There is evidence that the huntingtin protein hyperpolarizes the mitochondrial membrane and triggers neuronal apoptotic cell death.
Amyotrophic Lateral Sclerosis (ALS)
Amyotrophic lateral sclerosis is caused by cell death of the upper and lower motor neurons, which are responsible for controlling voluntary movements. The molecular pathological hallmark of ALS is the formation of inclusion bodies which is mainly composed of TAR DNA-binding protein 43 (TDP-43).
TDP-43 is a highly conserved nuclear protein that directly binds to DNA and regulates gene expression. Its pathological, mutated form relocates to the cytosol and forms protein aggregates.
Similarly, TDP-43 aggregates cause mitochondrial dysfunction. It is found that TDP-43 induces expression of pro-apoptotic proteins but there are also other reports indicating that TDP-43 leads to neuronal necrosis.
References:
- Brain Institute. (2020) ALS (Amyotrophic Lateral Sclerosis). Available from: https://www.ohsu.edu/brain-institute/als-amyotrophic-lateral-sclerosis.
- Chi, H., Chang, H. Y., & Sang, T. K. (2018). Neuronal Cell Death Mechanisms in Major Neurodegenerative Diseases. International journal of molecular sciences, 19(10), 3082. https://doi.org/10.3390/ijms19103082
- Elmore S. (2007). Apoptosis: a review of programmed cell death. Toxicologic pathology, 35(4), 495–516. https://doi.org/10.1080/01926230701320337
- Genetics Home Reference. (2020) Huntington Disease. Available from: https://ghr.nlm.nih.gov/condition/huntington-disease.
- Maji, S., K. (2020) Investigating the mechanism of Parkinson’s disease pathogenesis in test tube. Available from: https://rnd.iitb.ac.in/research-glimpse/investigating-mechanism-parkinsons-disease-pathogenesis-test-tube.
Further Reading
Last Updated: Aug 30, 2022