Sepsis can result when the body’s immune response to a specific infection gets out of control. Sepsis is a life-threatening disorder in which an overactive inflammatory reaction can instantly lead to multiple organ failure and even death.
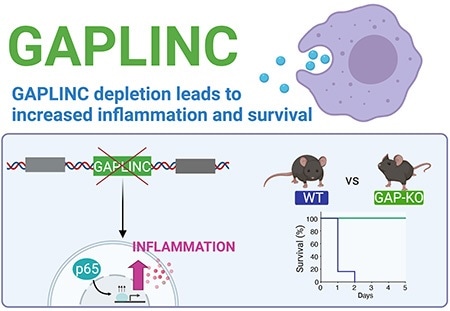
Reducing or eliminating the lncRNA GAPLINC led to enhanced expression of inflammatory genes in both mouse and human cells. In a mouse model of sepsis, knockout mice without GAPLINC were protected from endotoxic shock. Image Credit: Apple Vollmers.
Now, in a new research work, scientists from the University of California, Santa Cruz (UC Santa Cruz) have detected a long noncoding RNA, or lncRNA, molecule that controls the expression of pro-inflammatory genes in immune system cells, known as macrophages, and influences the vulnerability of mice to septic shock.
Called GAPLINC, the lncRNA was formerly investigated for its role in cancer, but this molecule turned out to be the most highly expressed lncRNA in macrophages, which has a core role to play in inflammation.
According to Susan Carpenter, assistant professor of molecular, cell and developmental biology at UC Santa Cruz, GAPLINC stood out when her laboratory conducted RNA sequencing of human macrophages as well as their precursors, that is, white blood cells known as monocytes.
Further experiments demonstrated that when GAPLINC is decreased or eliminated, inflammatory genes are highly expressed in both human and mouse cells. Paradoxically, this kiund of effect shielded mice from endotoxic shock and death in a sepsis mouse model.
The difference was significant, causing the death of all the normal (“wildtype”) within a day, whereas GAPLINC-knockout mice were able to survive.
Our hypothesis was that the knockout mice would do worse in a model of endotoxic shock, so we were surprised to find that they did much better.”
Susan Carpenter, Study Corresponding Author and Assistant Professor of Molecular, Cell and Developmental Biology, University of California, Santa Cruz
The study findings were published in the Proceedings of the National Academy of Sciences journal on February 1st, 2021.
Noncoding RNAs are essentially RNA molecules that are not translated into a protein but instead transcribed from the genome, and lncRNA is known to be the largest class of noncoding RNA.
In the recent past, investigators have discovered scores of lncRNAs in mammalian genomes that control the expression of genes in various biological processes. GAPLINC is one among the few examples of an lncRNA present in both mice and humans.
According to Apple Vollmers, a graduate student in Carpenter’s laboratory and the first author of the study, the RNA sequencing results of the team demonstrated that GAPLINC is overly expressed when monocytes differentiate into macrophages.
We thought initially that it was involved in regulating differentiation, but when we depleted it, that turned on inflammatory genes. It’s a low level of expression, so the genes are not turning on at the same level as when a macrophage is activated in response to a pathogen.”
Apple Vollmers, Study First Author and Graduate Student, University of California, Santa Cruz
Endotoxic shock
In the sepsis mouse model, mice were exposed to a component of gram-negative bacteria known as lipopolysaccharide (LPS, also called endotoxin), which typically stimulates septic shock in bacterial infections. Carpenter added that beginning from a low baseline level of inflammatory gene expression may make the inflammatory resection to LPS less of a shock to the system.
Instead of going from zero to 100, you might go from 10 to 100, and we think that provides some protection, but we’re not sure why.”
Susan Carpenter, Study Corresponding Author and Assistant Professor of Molecular, Cell and Developmental Biology, University of California - Santa Cruz
Macrophages are one of the first cells involved in the body’s reaction to an infection or injury. Macrophages are first responders and patrollers that circulate in the blood as monocytes and differentiate into macrophages that migrate to places where they are required to heal an injury or fight an infection.
Carpenter added, “They help turn on inflammation, but they also play an important role in turning it off.”
When monocytes differentiate into macrophages, the expression of GAPLINC gets activated and it is deactivated after the cells are subjected to LPS.
“Something interesting is happening between those steps, where somehow modulating the expression of GAPLINC can be protective,” Carpenter further stated.
The inflammatory response in sepsis becomes excess as cells discharge a deluge of inflammatory proteins known as cytokines in sepsis. Cytokines are generally produced in a cascade, as a single cytokine activates its target cells to produce more cytokines.
Unregulated production of cytokines is usually referred to as “cytokine storm.” A process like this also triggers clotting, subsequently allowing blood clots to inhibit the oxygen-carrying blood flow to vital organs, resulting in organ failure and even death. In addition, sepsis can be caused by viral infections and may be implicated in extreme COVID-19 cases.
Carpenter added, “The biggest problem with sepsis is that it happens so fast, and once it gets going there are no good treatment options.”
Deeper interoperation of the role of GAPLINC in regulating the inflammatory response and septic shock could pave the way for new drug developments to target sepsis.
“We are a long way from understanding how you would target this therapeutically, but at least we have identified a pathway to home in on,” Carpenter concluded.