The discovery of antibiotics is a significant milestone, which saved many lives. However, the widespread use has led to the steadfast evolution of highly resistant bacterial strains, which threaten to take humanity back to the starting point against infectious diseases.
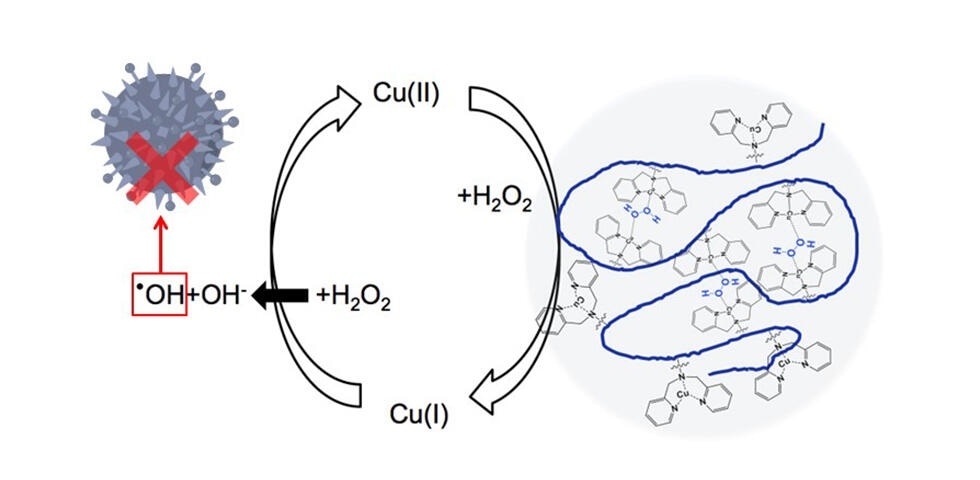
The proposed polymer, with its backbone shown in blue, creates regions with a high local density of copper side units (pendants). This helps reduce Cu(II) to Cu(I), the most difficult step in the redox reaction shown, to ultimately produce more hydroxyl radicals (•OH). Image Credit: Assistant Professor Shigehito Osawa.
Even when scientists are constantly on the lookout for new design concepts for antibacterial drugs, the overall development of new agents is on the decline.
To address this grave issue, researchers from the Tokyo University of Science, Japan, investigated a new approach to enhance the in vivo antibacterial activity of hydrogen peroxide (H2O2), a commonly used disinfectant.
A group of researchers headed by Assistant Professor Shigehito Osawa and Professor Hidenori Otsuka announced their success in improving the activity of H2O2 using carefully tailored copper-containing polymers. The research findings are published in the Macromolecular Rapid Communications journal.
Understanding the approach undertaken by the researchers helps identify how H2O2 acts against bacteria in the first place, and the role that copper plays. H2O2 can be decomposed into a hydroxyl radical (•OH) and a hydroxide anion (OH−), where the former is highly toxic to bacteria as it readily destroys certain biomolecules.
Copper in its first oxidation state, Cu(I), catalyzes the splitting of H2O2 into a hydroxyl radical and a hydroxide anion, turning into Cu(II) in the process through oxidation.
Inquisitively, H2O2 also catalyzes the reduction of Cu(II) to Cu(I), only if this reaction is mediated somehow. One way to facilitate this is to have Cu(II)-containing complexes get close enough together.
The only way that remains for Cu(II)-containing complexes dissolved in a solution to come close together is by accidentally bumping into each other. This involves an excessively high concentration of copper. The researchers identified a solution to this problem by taking inspiration from cellular chemistry.
In living organisms, copper forms complexes with proteins to efficiently catalyze redox reactions. For example, tyrosinase has two copper complex sites in close proximity to each other, which facilitates the formation of reaction intermediates between oxygen species and copper complexes. We thought we could leverage this type of mechanism in artificially produced polymers with copper complexes, even if dispersed in a solution.”
Shigehito Osawa, Assistant Professor, Tokyo University of Science
In light of the inspiration, the scientists created a long polymer chain with dipicolylamine (DPA) as copper-containing complexes. These DPA-copper complexes are attached to the long polymer backbone as “pendant groups.”
Upon dispersing the polymers in a solution, the Cu(II) atoms in the pendant groups are kept nearby and in locally high densities, which largely increases the chances that two of them would be close enough to be reduced to Cu(I) by H2O2.
The researchers conducted numerous experiments and demonstrated that the use of these tailored polymers resulted in greater catalytic activity for the splitting of H2O2, resulting in more OH• even for lesser concentrations of copper. Additional tests with Escherichia coli cultures demonstrated that these polymers highly enhance the antibacterial potential of H2O2.
Even when the study outcomes open up a new design avenue for antimicrobial drugs, there may be beneficial applications in the food industry too.
“Because copper is an essential nutrient for living organisms, the antibacterial agent developed in this study holds promise as an efficient food preservative, which could contribute to increasing the variety of foods that can be preserved over long shelf times.”
Shigehito Osawa, Assistant Professor, Tokyo University of Science
Hopefully, the new approach would help keep microscopic menaces at bay.
Source:
Journal reference:
Osawa, S., et al. (2021) Accelerated Redox Reaction of Hydrogen Peroxide by Employing Locally Concentrated State of Copper Catalysts on Polymer Chain. Macromolecular Rapid Communications. doi.org/10.1002/marc.202100274.