Using cutting-edge imaging methods on in vitro virus cells, researchers from the Institut Pasteur, CNRS, Vaccine Research Institute (VRI), and Université de Paris found a novel role of anti-HIV-1 antibodies. The researchers discovered that antibodies that have previously been shown to efficiently target the HIV-1 envelope (Env) protein can inhibit infected cells from releasing viral particles, thus curbing viral propagation.
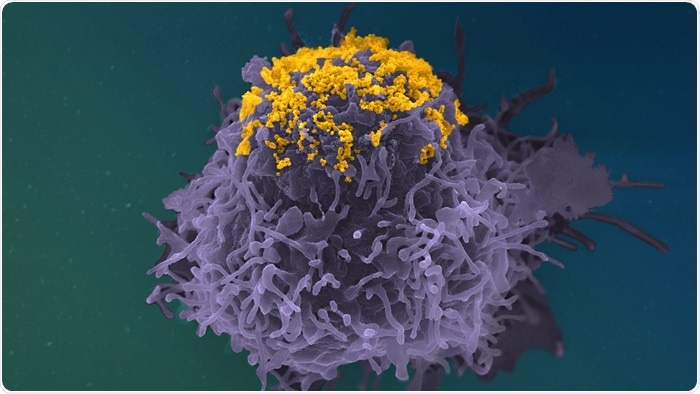
HIV particles (In Yellow) accumulating on the surface of an infected cell (In Purple). Colorized Scanning Electron Microscopy Image. Image Credit: © Stéphane Fremont, Jérémy Dufloo, Arnaud Echard, Timothée Bruel, Olivier Schwartz, Jean-Marc Panaud, Institut Pasteur
Because the antibodies are Y-shaped, they can bind between infected cells and viral particles or directly between viral particles. This chain of antibodies and viral particles blocks the transmission of the virus. These results show that, in addition to neutralization, these potent antibodies have a variety of antiviral actions. The work will be published on February 2nd, 2022, in the journal Nature Communications.
Broadly neutralizing antibodies (bNAbs) that target the viral envelope (Env) protein show a lot of promise for HIV-1 treatment. They were first discovered in a small number of people whose serum could block a variety of HIV strains. These antibodies have a variety of antiviral properties. They destroy infected cells as well as neutralize the virus, which prevents it from infecting new ones.
As a result, they are known as polyfunctional molecules. To improve the effectiveness of current antibodies or modify the selection criteria for new antibodies, it is vital to thoroughly comprehend the spectrum of these antiviral activities. Further research into the polyfunctionality of anti-HIV-1 antibodies is also beneficial to expand our understanding in the role of antibodies and therefore combat different viral infections.
Initially, researchers from the Institut Pasteur, the CNRS, the VRI, and the Université of Paris wanted to see if antibodies can stop infected cells from creating viral particles. To do so, scientists grew CD4 T cells (HIV's natural target) in vitro for 24 hours using different antibodies. The scientists, then, counted the number of viral particles generated by the cells in the culture media, as well as the number of viral particles still inside the cells.
The scientists were able to show that particular antibodies had increased the amount of virus in cells but decreased in the culture media as a consequence of their tests. This fascinating discovery led them to conclude that specific antibodies slowed the release of viral particles but did not stop them from being produced.
The scientists employed a variety of microscopy methods to study the creation of viral particles by cells to verify this notion. Fluorescence microscopy, a method for separating viral proteins, was employed to study the cells first. Researchers were able to show that infected cells collect huge amounts of mature viral protein as a result of this.
This discovery implies that cells collect complete virus particles. The scientists employed scanning electron microscopy to examine the surface of infected cells to pinpoint the precise position of the virus particles.
Using this method, we observed that these antibodies (bNAbs) prompt an accumulation of viral particles at the surface of cells, forming clusters and highly atypical structures.”
Timothée Bruel, Study Co-Last Author and Scientist, Virus and Immunity Unit, Institut Pasteur
The researchers then used immunogold labeling in conjunction with transmission electron microscopy. This allowed them to show that antibodies create a chain cluster by interposing themselves between virus particles and the infected cell. Experiments with mutant antibodies revealed that the antibodies’ Y shape is responsible for the clustered structure.
Their arms are strong enough to join two viruses or one virus to the infected cell membrane, and their attachment sites are powerful enough to cause this.
We have demonstrated that only the most powerful antibodies tether viral particles at the surface of infected cells. Trapped viral particles can no longer infect new cells.”
Olivier Schwartz, Study Co-Last Author and Head, Virus and Immunity Unit, Institut Pasteur
Source:
Journal reference:
Dufloo, J., et al. (2022) Broadly neutralizing anti-HIV-1 antibodies tether viral particles at the surface of infected cells. Nature Communications. doi.org/10.1038/s41467-022-28307-7.