As part of our SLAS Europe 2022 coverage, we speak to Jan Lichtenberg, vice president of SLAS, about the role Europe plays within life sciences research and how a new European start-up can stand out in the sector.
Please could you introduce yourself and tell us about your role at the Society for Laboratory Automation and Screening (SLAS)?
My name is Jan Lichtenburg, and I am currently the vice president of SLAS. I have been involved with SLAS since its very beginning as an exhibitor, contributor of scientific content, or a short course instructor.
I am an electrical and microtechnology engineer by training; I come from the microfluidic side, but I live and work in the life science space. It is the interdisciplinary atmosphere that SLAS brings to the table that has always been fascinating to me. When I was asked two years ago to be part of the board of directors of SLAS, I was honored. I served as a secretary for a year, the vice president this year, and then will be president next year.
It is a great organization to be part of, because bringing the scientific community and the vendor community together, is so important for our industry. At the end of the day, the innovation that we want to accomplish only happens if we have vendors and users on board.
Introducing InSphero
Alongside your role at SLAS, you are also the CEO and Founder of InSphero, a company helping to accelerate drug discovery and further our understanding of disease. What made you decide to start a company in this space, and could you tell us about some of InSphero's core values and missions?
We started InSphero with the idea to bring the human patient back into the center of drug discovery and development. Currently, that is not the case: Starting from a hypothesis the organizations identify their targets and take them through the whole development chain. Finally, at the very last minute, they begin working with patients.
From a safety perspective, this process makes sense. From an efficacy perspective, it does not.
The human patient must be the beginning and the end of the drug discovery process. This is what our organization is focusing on. We are rebuilding parts of the human patient - tissues and organs - in a highly scalable fashion in the lab to use them for safety and efficacy testing. We have liver tissues, heart muscle tissue, brain tissue, pancreatic tissue, and even skin.
These human tissue models play relevant roles throughout the R&D process: to see whether a drug works, whether there are off-target effects, or to address safety concerns, all of which need to be mitigated early on. You can do this with our technology in a highly predictable way in the lab.
Ultimately, this accelerates drug discovery. It also enables drug discovery for some diseases that are not currently treated. We also hope to reduce animal testing, as we not only get better at rebuilding the human in the lab but also need for human in-models for testing modern drug modalities that are not working correctly in the animal.
This is the mission that we started with. We wrote our business plan in 2008, submitted it for the first business plan competitions in 2009, and then had Novartis as the first customer in the same year. This was also when we left our old jobs and started the company with three founders.
Our technology was mature coming out of the University of Zurich in Switzerland and ETH. At the same time, there was a growing need in the pharmaceutical industry for novel methodologies in this area.
Akura™ 96 Spheroid Microplate | All-in-one plates for spheroids and organoids
You participated in a panel session at SLAS EU titled 'Perspective in the European Life Science Ecosystem.' Can you tell us more about what was discussed in this session and the importance of highlighting Europe as a major player within the life sciences community?
This was a great session, and I think it was also a good for the SLAS Europe conference to embrace the specifics of the European market. It is not just a copy of SLAS International in the US. We had a great panel of larger and smaller start-up companies, as well as entrepreneurs that have successfully sold their companies or even brought them to the stock exchange via an IPO.
The big question is: "What made you successful in the European ecosystem?". It is important to assess what may be easier or more difficult here, and it was interesting to hear this from the different groups.
We are also concerned with growing the company not solely within Europe but also outside Europe: approximately 50% of the R&D market in the pharmaceutical industry is in the US. Our organization cannot stay in Europe alone; we need to take things international quickly.
This can also be seen when working with larger pharma companies; often, they have their research centers in Basel, San Francisco, and Boston. It is crucial to align the interest of these stakeholders. I think this is something that we have all struggled with, and we all managed in different ways.
Europe is home to 43 of the World's top 100 universities for life sciences and publishes roughly twice as many scientific articles as the US. As well as this, what else makes Europe an exciting place to be within the life sciences community, and what more can be done regarding investment to help continue its development?
Firstly, I think the numbers speak for themselves, both in terms of scientific quality and output – meaning that Europe is a great place for science. We see this in terms of patents that are filed as well as in terms of articles that are published. We also see this in terms of European labs' innovation. Commercial mRNA vaccines, which were so important during the pandemic, are just one example where Europe has excelled.
Now, at the same time, Europe remains a pretty fragmented environment. We have a lot of states, cultures, and languages. We have a lot of different regulations despite the encompassing European Union. We have a lot of different funding systems for start-up companies. We have different tax regulations. So, all things considered, it is a bit more challenging to navigate.
My company is based in Switzerland, technically within Europe but not part of the European Union. This means that we have a different set of advantages and disadvantages. We are very close to fantastic universities, the University of Zurich, ETH, and the University of Basel, to name a few academic powerhouses. We have some fantastic large pharmaceutical companies in Switzerland, which are very strong in research, as well as many small innovators. So, this range opens a lot of opportunities for interactions.
We have a good tax environment and flexible labor laws, which makes it easy for small companies to hire because they know that they can also scale back if necessary, which sometimes, unfortunately, is necessary for survival. There are unique advantages if you can navigate them.
There are also disadvantages. In the early phase of a start-up, countries like Switzerland do not provide non-dilutive funding. They support academic projects that can help you innovate, but no direct financial support. This is, therefore, a bit of a disadvantage for Swiss start-ups compared to others in Europe.
Generally speaking, Europe as a whole has a bit of a translation problem from science into technology and then into products and services. Americans tend to be much better at bringing out the big guns when technology has proven its usefulness and needs to be scaled up so that it can be introduced into the market. Unfortunately, it is sometimes seen that American start-ups often overtake European ones or that Europeans move over to the US to become more successful there. I believe that is something that we need to revise.

Image Credit: sdecoret/Shutterstock.com
Biotechnology is a field with the potential to revolutionize many sectors within the life sciences, including diagnostics, agricultural science, and drug discovery. Therefore, in 2018, the European bioeconomy strategy was published to help 'speed up the progression towards a European bioeconomy.' How important is this strategy to European life sciences companies such as InSphero, and how can having a strategy in place help strengthen Europe's place within science while also contributing to the Paris Agreement and the Sustainable Development Goals (SDGs)?
The European bio-economy strategy is very much focused on natural resources and on sustainable development goals, which is extremely valuable for the European market. It is not considered a major game-changer for our particular industry because the key goals are to make better use of natural resources, tackle climate change, and generally be more sustainable as a society.
However, the implications are important for us as a company and are part of our core values. Biotech is not a very green technology. You use a lot of water as well as energy to filter air and run air condition your systems. We have a lot of waste because we rely so much on products that can only be used once. So, these are aspects that enables us to contribute to SDGs.
For example, we are currently addressing measures to reduce CO2 emissions by reducing animal testing. Animal testing produces a great amount of CO2 and requires a lot of water. Each of the cages in a rodent facility has its own conditioned air supply, for instance. So, by moving to in vitro testing, we can make a meaningful contribution to reducing CO2 emissions.
I think there are a lot of technologies, like CRISPR, which can have a significant impact on food supply, as well as a reduction in the use of pesticides and water if you have a crop that is optimized for a specific climate. This is not an area in which we can be particularly helpful as InSphero, but we can make our contributions by being more sustainable in how we act every day.
As our world is fast-evolving with scientific advancements being made at great speeds, we are seeing many innovative new start-up companies emerging, especially within Europe. What are some of the challenges new businesses need to consider when starting in Europe, and what advice would you give to a new start-up looking to leverage the European ecosystem to help build their global business?
The main start-up challenges today are the same as they were ten years ago: for instance, considering access to funding. However, students – particularly postdocs that are ready to take their technology into a spinoff - are so much better educated in entrepreneurship today than we were 15 years ago. The way they present the business plans, and their whole entrepreneurial acumen, is amazing. I think a lot has improved.
However, it also makes the competition for funding and the competition in the market more difficult, which I believe is a good thing: almost a survival-of-the-fittest situation. The most relevant risk for every start-up company is to develop something that nobody needs. We all love the science we are carrying out in the lab and want to take it to the market. We believe that other people will see the same beauty in it and will also use it, but that is not necessarily the case.
Often, we come across groundbreaking technology looking for an application, which is not how things should be. The need should exist first, and then a solution should be developed to fulfill it.Organizations that operate like that are the most successful start-ups.
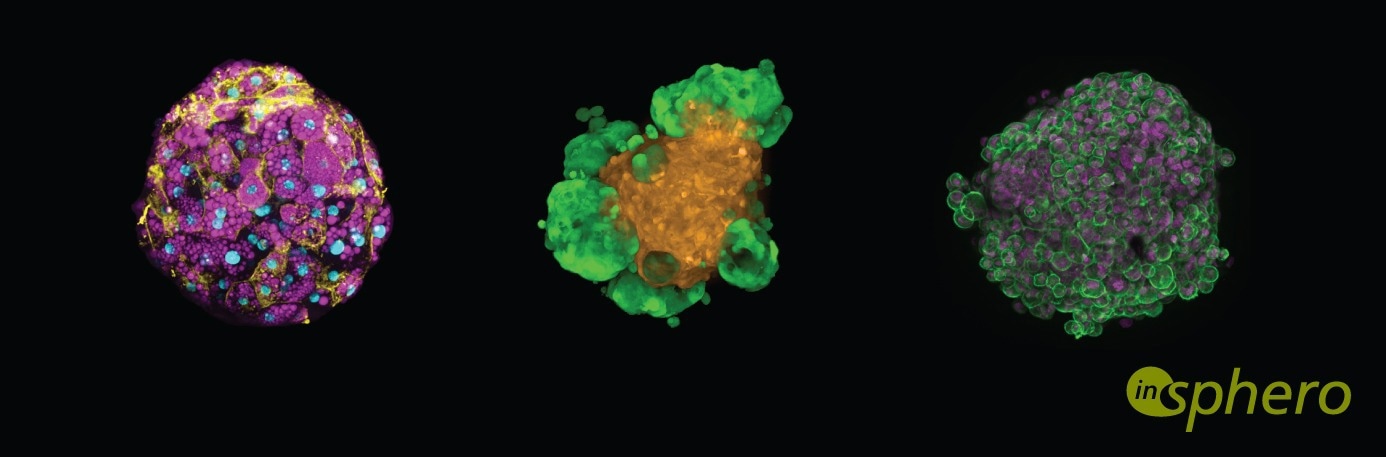
Image Credit: InSphero
When starting a new business in the biomedical and life sciences sector, you often compete against well-established pharmaceutical companies. How can a new European company stand out from pharma companies and create its own space in the market?
The 'war for talents' is definitely fierce. A start-up, typically, cannot compete on pay or fringe benefits. You can compete, however, in terms of purpose and ownership in the company. The purpose is something that we see is becoming so much more relevant for the younger folks that start in our company. The younger generation is well-educated, but they want to understand why they come to work every morning, what they can contribute and how their contribution fits into the bigger scheme of things.
A smaller company can be better at finding its mission and purpose than a large company because they are less dependent on published quarterly KPIs and investor needs. Many people are driven to join companies like ours because they like the purpose and can play a more significant role in a smaller system. This means that they take on more responsibilities earlier on. They are not simply fulfilling a small task in a bigger process but instead actually having to take things from the lab to the customer.
That is extremely exciting for employees. We see people moving from big pharma to companies like ours because they want to start afresh. To us, this is great because it adds diversity to the company. This means that we have different age groups, which definitely helps to add life to our approach.

Image Credit: Andrii Yalanskyi/Shutterstock.com
The COVID-19 pandemic has taught us many lessons, especially the importance of communication and collaboration. How important do you believe these skills are for the success of life sciences companies? How does having good communication skills not only benefit future partnerships but customer relationships also?
Communication - being able to tell your story – is essential, but we also think that being able to listen to what the other side says is super important. When discussing communication skills, we often talk about 'sending', speaking, or directly communicating to an employee. Instead, when we talk to customers, we primarily want to listen – which is not always easy because we are not used to this. Listening to what customers are working on, what their needs are, listening to both the issues they have solved and the issues they have not solved. That sometimes requires us to dial back in our need to send all the time.
During the pandemic, we had many virtual meetings, but we lost the scientific debate, the disagreement, and the challenge on certain things. The intersection of debate is where science gets better: you present your research, and people challenge you, and you defend yourself, or you get some great ideas, but both sides learn, and both sides leave the conversation with a gain.
That is something that I have not really seen a lot in virtual meetings because it is easy to just listen to a presentation. The drawback is that you do not engage in the discussion, which is a problem.
We are also seeing smaller European countries receive increased attention within the life sciences. How can European nations work together to support smaller regions and countries to become established powers in the field of life sciences?
In my opinion, this collaboration already works extremely well, particularly when you consider countries like Denmark and Austria.
I think the European research funding landscape helps empower these small countries to participate in bigger networks. That is extremely important. Large countries, like Germany, France, and the Netherlands, have their national funding schemes, which is great. True innovation happens if you bring together different European countries and have an excellent geographic balance. It is essential to have a good balance between small players and large players. The European Union makes that collaboration a prerequisite, which is essential, and receiving funding is vital to help these smaller countries stay in the game.
What do you believe the future of life sciences research in Europe to look like? Are you hopeful that we will see Europe becoming more prominent in breakthrough discoveries with continued innovation, funding, and collaboration between companies?
I have a very positive outlook on the future of European life sciences. I think, first of all, we remain a highly science-based and fact-focused part of the world. This is not true for every leading global country: in some, there may be a concern - and even a growing disrespect for – science, which is a big problem. Though it happens also on the fringes of Europe, it does seem to be mostly under control.
Efforts like the European Investment Council not only support science but also help to bridge the funding gap: allowing companies to stand on their own feet before they are ready to be investible for larger investment funds. I think that is a good move. It is also great to see American investors and investors from Asia taking more interest in European companies and establishing offices to invest in them. There are a lot of moving parts that are driving this in the right direction.

Image Credit: watchara/Shutterstock.com
Are there any particular fields within the life sciences you are particularly excited to watch evolve over the coming years? What should people be watching out for?
There are three main trends. The first of these is precision medicine – otherwise known as personalized medicine. It is essential to get a better understanding of patient diversity and the fact that patients react differently to different diseases, as well as to different medications.
It is essential to better understand which drug works for a patient and which does not. I anticipate that we will see a great deal of innovation in that space, which will drive drug discovery and application into a whole new area.
The second important aspect is gene editing, both as a research and therapeutic tool. I fully support great technology, but we need to invest into engaging with the end-user about the concerns that they about gene editing. If we are successful, this can be a powerful tool: not just in medical and therapeutic applications but also in the arena of natural resources, food supply, and so on.
The last aspect is big data and artificial intelligence. A lot of the technologies that we develop create big data sets. We work a lot with imaging and next-gen sequencing. If you do not have the bioinformatics tools to help you condense that information into a nucleus that allows you to make a decision, all that information is worthless. Bringing that all together is fantastic.
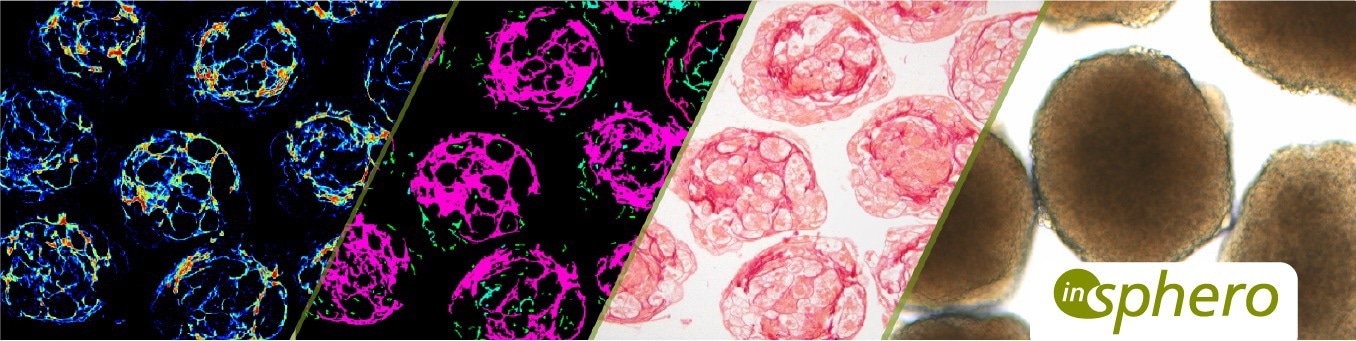
Image Credit: InSphero
About Jan Lichtenberg
Jan Lichtenberg, Ph.D., is Co-Founder and CEO of Swiss- and US-based InSphero Inc., the largest biotech specialized in 3D cell-culture technologies for discovery and safety. InSphero’s patented assay-ready 3D microtissues mimic the structure and functionality of organ tissue, e.g. liver, pancreas, or tumors including disease states like diabetes or NASH. The 3D microtissues allow for more predictive and reliable compound profiling for discovery and safety testing in a highly robust, rapid and cost-efficient way.
Jan co-founded InSphero in 2009 and grew the company to 65 employees in Switzerland and the US while expanding the business to encompass all top 15 global pharmaceutical companies. Prior to InSphero, Jan had VP R&D and Product Management positions at Hocoma AG (medical robotics) and Uwatec (microelectronics). He holds a Ph.D. from the University of Neuchâtel and managed a research group at the Swiss Federal Institute of Technology (ETH), Zurich. Since 2021 he is a Board Member of the Society of Laboratory Science and Screening (SLAS).
About InSphero
InSphero is the pioneer of industrial-grade, 3D-cell-based assay solutions and scaffold-free 3D organ-on-a-chip technology. Through partnerships, InSphero supports pharmaceutical and biotechnology researchers in successful decision-making by accurately rebuilding the human physiology in vitro. Its robust and precisely engineered suite of 3D InSight™ human tissue platforms are used by major pharmaceutical companies worldwide to increase efficiency in drug discovery and safety testing.
The company specializes in liver toxicology, metabolic diseases (e.g., T1 & T2 diabetes and NAFLD & NASH liver disease), and oncology (with a focus on immuno-oncology and PDX models). The scalable Akura™ technology underlying the company’s 3D InSight™ Discovery and Safety Platforms includes 96 and 384-well plate formats and the Akura™ Flow organ-on-a-chip system to drive efficient innovation throughout all phases of drug development.