According to a recent study, brain cells with the same “birthdate” are more likely to connect into cooperative signaling circuits that perform a variety of tasks, including memory storage.
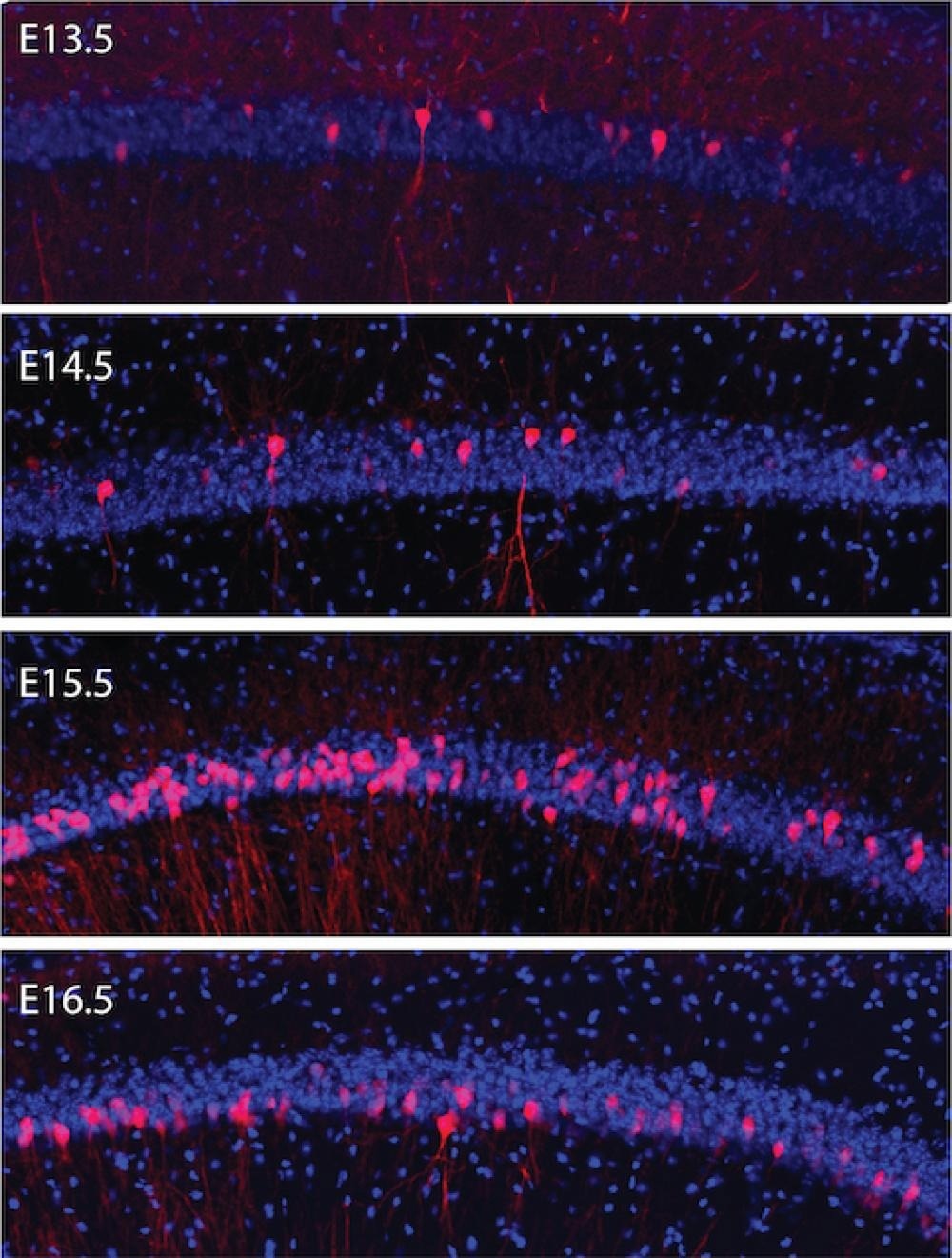 Fluorescence microscopy shows groups of brain cells that are born together on the same days (pink) and different days (blue) in the hippocampus of a mouse embryo. Many more neurons are born on embryonic day 15.5 (E15.5) than on earlier or later days. The study found that cells with the same birthdates during development form circuits that work together to encode similar memories. Image Credit: Roman Huszár et al., Nature Neuroscience.
Fluorescence microscopy shows groups of brain cells that are born together on the same days (pink) and different days (blue) in the hippocampus of a mouse embryo. Many more neurons are born on embryonic day 15.5 (E15.5) than on earlier or later days. The study found that cells with the same birthdates during development form circuits that work together to encode similar memories. Image Credit: Roman Huszár et al., Nature Neuroscience.
The new study on the growing brains of mice led by scientists from NYU Grossman School of Medicine discovered that brain cells (neurons) with the same birthdate displayed distinctive connectivity and activity throughout the animals' adult lives, whether they were asleep or awake.
The results, which were published on August 22nd, 2022, in Nature Neuroscience, imply that evolution used the organized birth of neurons—by gestational day—to create specialized microcircuits in the hippocampus, the part of the brain responsible for memory formation.
The researchers hypothesize that the brain may make use of the neuronal layers’ gradual development to generate neural templates, or “Lego parts,” that connect each new event to an old template as it is remembered.
According to the scientists, these principles of circuit building would imply that cells that are born together are more likely to encode memories together and to perish together, suggesting a possible link between neuronal birthdate and disorders like autism and Alzheimer’s disease. The developing brain may be more susceptible to viral infections, toxins, or alcohol on specific gestational days due to variations in the number of cells born on different days.
Our study’s results suggest that which day a hippocampal neuron is born strongly influences both how that single cell performs, and how populations of such cells signal together throughout life.”
György Buzsáki MD, PhD, Study Senior Author and the Biggs Professor, Department of Neuroscience and Physiology, NYU Langone Health
“This work may reshape how we study neurodevelopmental disorders, which have traditionally been looked at through a molecular or genetic, rather than a developmental, lens,” states Dr Buzsáki, also a member of the Neuroscience Institute at NYU Langone.
New understanding of memory storage
The novel aspect of the current study is the ability to follow the neuronal activity of a specific birthdate into adulthood. The researchers used a method that allowed them to insert DNA into cells that were dividing into neurons in the womb to achieve this.
Similar to a barcode, the DNA expressed markers that identified brain cells that were born on the same day. The researchers were subsequently able to analyze these neurons in the adult animal using this labeling technique.
The latest research discovered that neurons with the same birthdate had a propensity to “co-fire” together, which is characterized by coordinated swings in their positive and negative charges and enables them to convey electrical messages jointly. The scientists speculate that the co-firing may be caused by shared synapses connecting neurons with the same birthdate.
Studies in the past had demonstrated that patterns of combined neuronal activity during waking and sleeping can be used to define activity in the hippocampus. The “sharp wave-ripple,” a cyclical burst of activity that occurs during sleep when daily memories are organized for long-term memory storage, is named after the shape it takes when graphically captured by EEG, a device that captures brain activity.
Our results show that neurons born on the same day become part of the same cooperating assemblies, and participate in the same sharp wave-ripples and represent the same memories. These relationships, and the pre-set templates they encode, have a key implication for hippocampal function: the storage of a memory about a place or event.”
Roman Huszár, Study First Author and Graduate Student, NYU Langone Health
Roman Huszár is a student in Dr. Buzsáki’s lab.
The researchers will conduct more research in the future to determine the genes involved in behavior and memory development in neurons with the same birthdate located in various brain areas.
Source:
Journal reference:
Huszár, R., et al. (2022) Preconfigured dynamics in the hippocampus are guided by embryonic birthdate and rate of neurogenesis. Nature Neuroscience. doi.org/10.1038/s41593-022-01138-x.