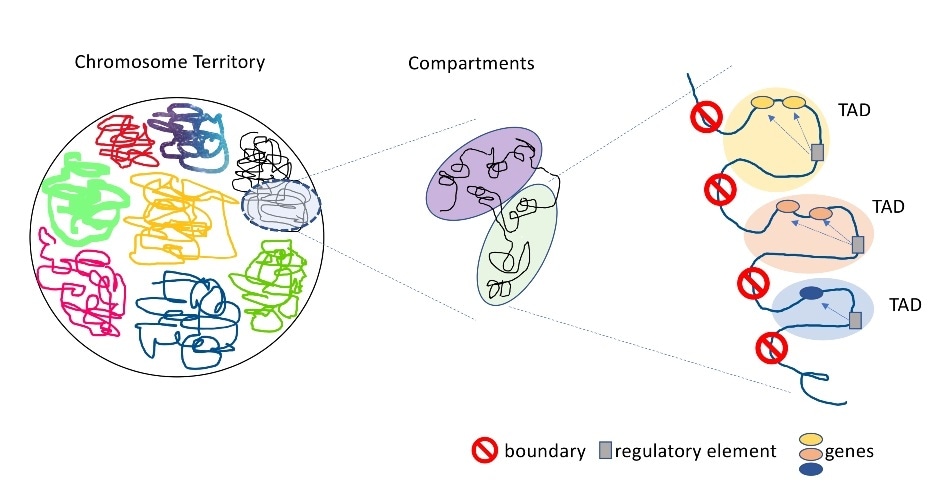
Topologically associated domains (TADs) are distinct regions of the genome with strict boundaries that keep the domains from interacting with genetic material in neighboring TADs. Image Credit: Jin Szatkiewicz, PhD, UNC School of Medicine.
The study, which was jointly headed by senior author Jin Szatkiewicz, Ph.D., associate professor in the UNC Department of Genetics, implies that rare structural genetic variants could be involved in schizophrenia. The study was published in the Nature Communications journal.
The co-senior author of the study is Patrick Sullivan, MD, the Yeargen Distinguished Professor of Psychiatry and Genetics at the UNC School of Medicine and Director of the Center for Psychiatric Genomics.
Our results suggest that ultra-rare structural variants that affect the boundaries of a specific genome structure increase risk for schizophrenia. Alterations in these boundaries may lead to dysregulation of gene expression, and we think future mechanistic studies could determine the precise functional effects these variants have on biology.”
Jin Szatkiewicz, Study Senior Author and Associate Professor, Department of Genetics, University of North Carolina School of Medicine
Earlier studies on the genetics of schizophrenia mainly involved the use of common genetic variations called SNPs (changes in common genetic sequences and each impacting a single nucleotide), rare changes in the part of DNA that give instructions for developing proteins, or very large structural changes (variations that impact a few hundred thousands of nucleotides).
Such studies give some specifics of the genome, but render a large portion of the genome quite mysterious, as it possibly associates with schizophrenia.
Using a method known as whole genome sequencing (WGS), Szatkiewicz and collaborators have analyzed the whole genome as reported in the Nature Communications study. WGS is not used extensively because it is very costly.
For this research, international collaboration was formed by pooling funds from the National Institute of Mental Health grants and matching funds from Sweden-based SciLife Labs.
The aim was to perform deep whole-genome sequencing on 1,165 people suffering from schizophrenia and 1,000 controls—the largest known WGS study of schizophrenia so far.
Consequently, new findings were made. For example, researchers identified previously undetectable mutations in DNA which they had never observed in schizophrenia before.
Specifically, this research emphasized the potential role played by a three-dimensional (3D) genome structure called topologically associated domains (TADs) in the development of schizophrenia.
TADs are distinct regions of the genome with stringent boundaries between them that restrict the domains from communicating with genetic material in adjacent TADs. Breaking or shifting these boundaries enables interactions between genes and regulatory elements that generally would not interact.
When these interactions take place, gene expression could alter in undesirable ways that can potentially lead to the development of cancers, congenital defects, and developmental disorders.
This research discovered that very rare structural variants impacting the brain’s TAD boundaries occur considerably more often in people suffering from schizophrenia than in individuals without it.
Structural variants can be defined as large mutations that may involve sequences that are not in the typical genome or involve duplicated or missing genetic sequences. This outcome indicates that missing or misplaced TAD boundaries may also play a role in the development of schizophrenia.
This research was also the first to identify the association between TAD anomalies and the progression of schizophrenia.
This study has emphasized how TADs-affecting structural variants are major candidates for more mechanistic researches on the biology of schizophrenia.
A possible future investigation would be to work with patient-derived cells with these TADs-affecting mutations and figure out what exactly happened at the molecular level. In the future, we could use this information about the TAD effects to help develop drugs or precision medicine treatments that could repair disrupted TADs or affected gene expressions which may improve patient outcomes.”
Jin Szatkiewicz, Study Senior Author and Associate Professor, Department of Genetics, University of North Carolina School of Medicine
Szatkiewicz is also an adjunct assistant professor of psychiatry at UNC.
The research will be merged with other WGS studies to increase the size of the sample, to additionally validate these outcomes. It will also support the scientific community to build on the evolving genetic mysteries of schizophrenia.
Source:
Journal reference:
Halvorsen, M., et al. (2020) Increased burden of ultra-rare structural variants localizing to boundaries of topologically associated domains in schizophrenia. Nature Communications. doi.org/10.1038/s41467-020-15707-w.