In this interview, AZoLifeSciences talks to Dr. Shane Liddelow about the research in his lab into neurodegenerative diseases like glaucoma, and the theory that astrocytes are the crucial perpetrators of these diseases, a phenomenon which could be exploited in future therapies.
What led you to begin this research?
We have been interested in the role of reactive astrocytes and how different subtypes may interact with other cells in the brain for several years. While we have previously worked on these interactions in isolated cells in a culture dish, we were always keen to determine how well these functions transferred to an in vivo system.
Our initial discoveries suggested that a subset of reactive astrocytes could drive the death of neurons in a dish, however, we also knew that these reactive astrocytes were present in regions of the brain where neurons were not dying – we were intrigued why this was and guessed that perhaps neurons had to be damaged to be removed.
The visual system provides a fantastically accessible model to compare an acute injury (axotomy of retinal neurons) with a chronic degeneration model (bead-occlusion model of glaucoma) for us to investigate different components of theseastrocyte-neuron interactions.
.jpg)
Image Credit: Kateryna Kon/Shutterstock.com
What is the ‘reactive’ phenomenon your study has found regarding brain cells and nerves?
Following infection, injury, disease, and other pathological conditions, astrocytes respond by becoming ‘reactive’ in a process known as ‘gliosis’. There are many different types of reactive astrocytes – each with their own characteristic gene expression profiles, protein levels, and functional changes from ‘normal’ physiological astrocytes.
We previously reported that upon inflammatory insult, microglia activate a subtype of reactive astrocytes (Nature, 2017, 541:481-487). Here we showed that this subset of reactive astrocytes is likely driving the death of neurons by an as-yet-unidentified secreted molecule. This is in contrast to several other forms of reactive astrocytes that are reported to have beneficial roles following neuron injury, or other non-neurotoxic functions.
In fact, the advent of single-cell transcriptomics is now highlighting a very diverse array of possible reactive astrocyte substrates – a very exciting time for research in astrocyte biology.
What is glaucoma and how could it be driven by this phenomenon?
Glaucoma is a chronic progressive neurodegenerative disease that affects the eye. In patients with glaucoma, there are characteristic increases in the pressure within the eye (intraocular pressure), death of neurons, and progressive loss of vision.
We hypothesized that the death of these neurons was due to reactive astrocytes that are formed as a result of an immune response to increases in intraocular pressure, rather than as a result of the pressure itself.
Image Credit: ALPA PROD/Shutterstock.com
What other neurodegenerative diseases could be driven by this phenomenon?
Along with many amazing collaborators, we have previously reported that these reactive astrocytes are present in mouse models of (among other diseases) Alzheimer’s disease (Shi et al., 2017 Nature 549:523-527), Parkinson’s disease (Yun et al., 2018 Nature Medicine 24(7):931–938), oligodendroglioma (a cancer of oligodendrocytes, another important cell in the brain, Gibson et al., 2019, Cell 176(1-2):43-55), and most recently in human astrocytes, derived from patients with Alzheimer’s disease (Barbar et al., 2020, Neuron S0896-6273:30360-3).
It is important to note however that while we see this subset of reactive astrocytes in all these disease models, they are not the only form that is present. They are only present in regions of degeneration in the brains of affected animals and human patients, and overall we estimate they represent only a few percent of all astrocytes across the brain and spinal cord.
How did you use rodent models to show this theory?
A graduate student, Kevin Guttenplan used our acute (axon injury) and chronic (glaucoma) model of eye injury to show that the neuron-killing astrocytes are present rapidly following injury/disease, and they persist for several weeks. He also showed that using a global knock out mouse model that is unable to produce these astrocytes there is no death of retinal neurons in either model.
While this does not remove the possibility that other cells (e.g. immune cells like microglia) could also be playing a role in this neuron death model, we have previously shown that in the absence of reactive astrocytes the immune response is quite similar – suggesting they are likely not the active triggers of neuron death.
Kevin also used antibodies that neutralize factors that cause the formation of these neurotoxic reactive astrocytes – to offset the problems associated with using a global knockout mouse.
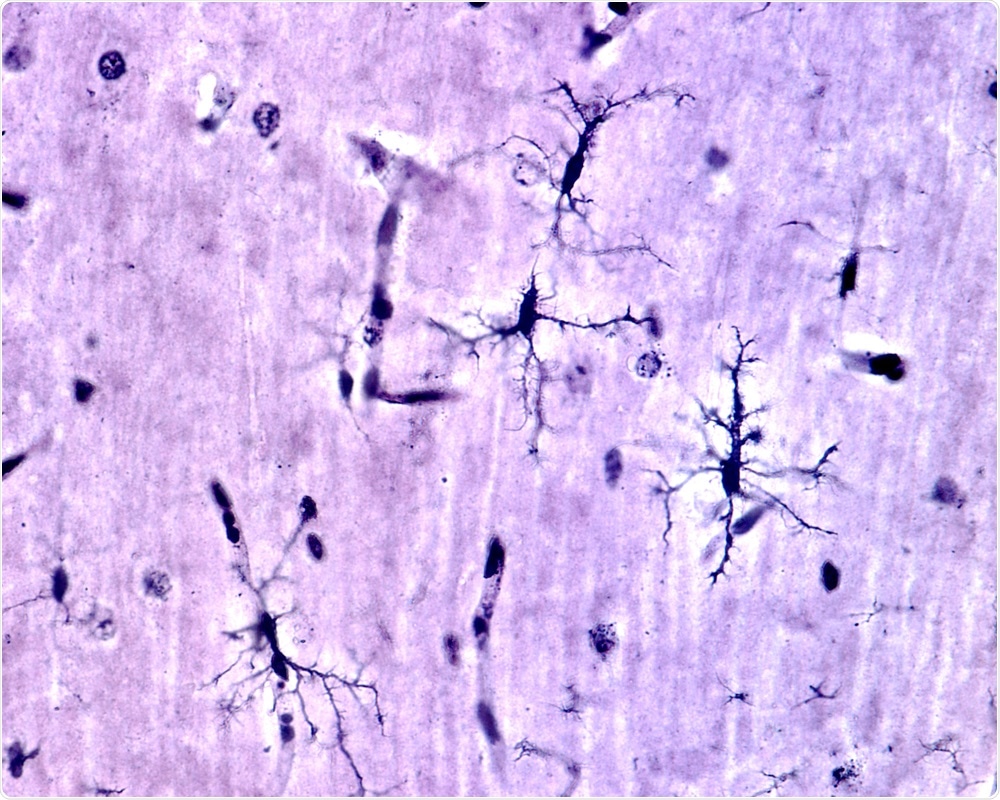
Image Credit: Jose Luis Calvo/Shutterstock.com
How could this theory be used to develop new ways to treat glaucoma and other neurodegenerative diseases?
Current therapies largely focus on neuron-specific targets, or on the modulation of the immune system. Astrocyte are likely situated between these two cell types in the neuron-death axis.
Astrocytes also present a very large, stable target: they are a major cell population in the brain (estimates range up to 50% of all cells in some brain regions), and they are static – that is they do not move around the brain, or enter from the periphery, like immune cells and thus provide an easier therapeutic target.
By combining astrocyte therapies that stop driving the death of neurons, together with immunotherapies to control inflammation, and therapies to promote neuron regeneration and function, we feel we have a real chance at treating glaucoma, and a wide range of other neurodegenerative diseases.
What has this study indicated about future research in this area and where focusses should lie?
This study had two exciting highlights: 1) astrocytes are only able to kill neurons that are already damaged (that is, there is no innocent bystander effect); and 2) if protected from death, neurons that remain still have some electrical activity (suggesting they could still play an important role in the recovery from disease and injury).
The first of these results, that astrocytes only kill damaged neurons, makes sense, as we do not see wide-spread death of neurons even when these reactive astrocytes are present. It seems that there is an important decision made by the system to remove damaged or dysfunctional neurons in an attempt to preserve functional neuronal networks in the brain as a whole.
But, having said that, this seems to be a short-term hurdle, and if neurons can be preserved beyond this initial damaging situation, we may be able to coax them back to normal function. This is a very exciting prospect for the development of future therapies.
What are the next steps for your research?
We remain very interested in how different subsets of reactive astrocytes change their interactions with other cells in the brain and spinal cord following injury/infection and during disease.
Most recently, we have published the role of these astrocytes in amyotrophic lateral sclerosis – showing they are important mediators of the progression of neurodegeneration in this disease. By blocking formation of neurotoxic reactive astrocytes, we dramatically slow the progression of the disease.
What is the identity of the astrocyte-derived neurotoxin? At what stage during disease are these, or other reactive astrocytes, present? And can we modulate astrocyte reactive states to help treat chronic neurodegenerative disease? These are big questions that our lab is actively pursuing.
Where can readers find more information?
Our lab website – www.liddelowlab.com – has links to our most recent research, published articles, and current news.
About Dr. Shane Liddelow

Shane Liddelow is an Assistant Professor of Neuroscience & Physiology and Ophthalmology, at the Neuroscience Institute at NYU Grossman School of Medicine in New York City. He trained in pharmacology, neuroscience, and glial biology at the University of Melbourne, Australia, and at Stanford University.
His lab is interested in understanding the molecular pathways and mediators that drive astrocyte reactive states in neurodegenerative disease.
In 2020 the UK’s leading dementia research charity, Alzheimer’s Research UK, awarded Dr. Liddelow the prestigious David Hague Early Career Investigator of the Year Award, and in 2019 the Alzheimer’s Association presented its Inge Grundke-Iqbal Award for Alzheimer’s Research in recognition of his work as the most impactful study published in Alzheimer’s research during the previous two calendar years.
His lab is funded by NYU School of Medicine, the Cure Alzheimer’s Fund, the National Institutes of Health, The Blas Frangione Foundation, MD Anderson Neurodegenerative Disease Consortium, and generous Anonymous Donors.
His work has been published in Nature, Science, Cell, and other leading journals. He is an advocate of under-represented minorities and immigrants in science and is passionate about helping to bridge the gap between research and public understanding of science.