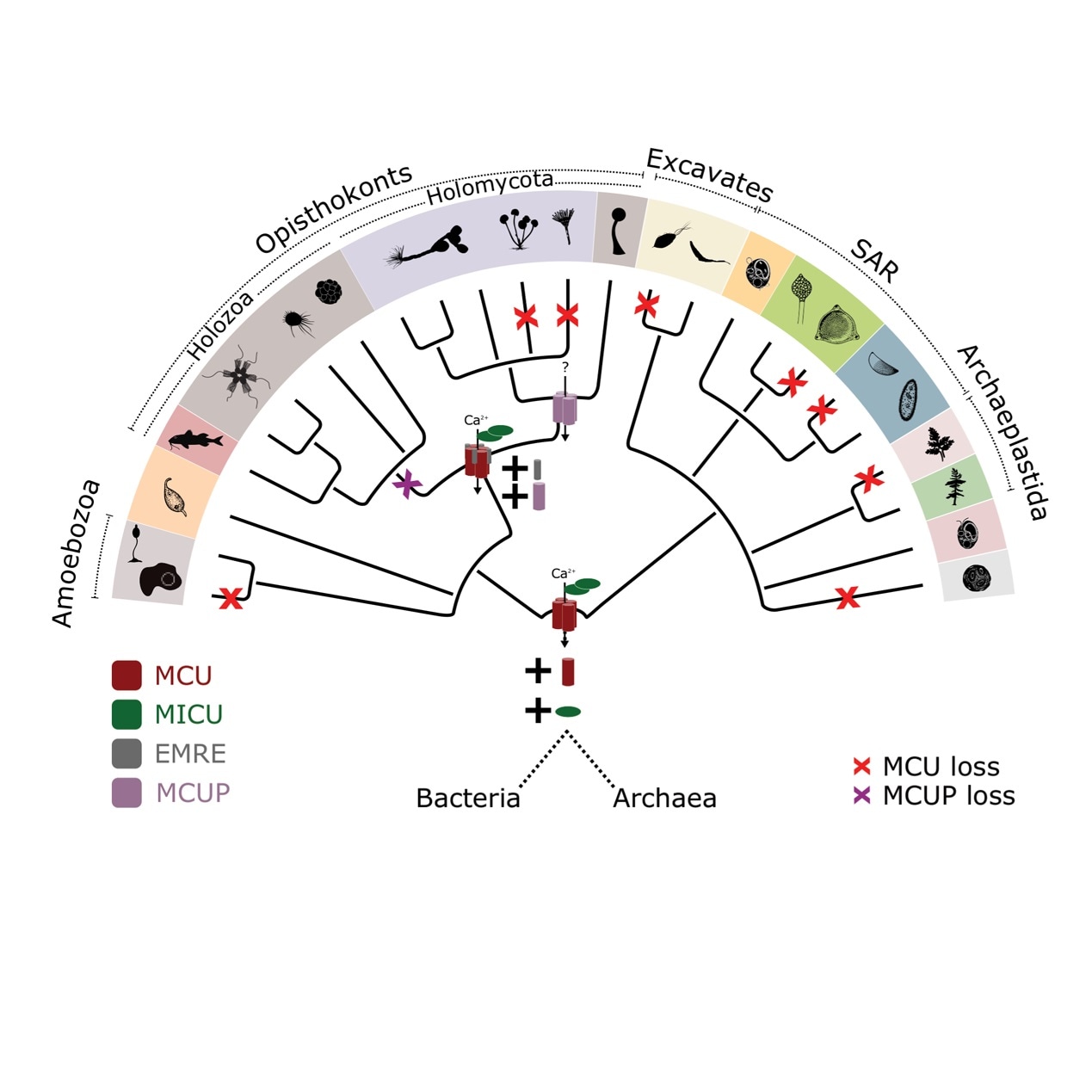
According to a study performed by the Comparative Genomics Laboratory at the Institute for Research in Biomedicine (IRB Barcelona), the system that controls calcium levels in cells duplicated, giving rise to a pair of non-equivalent systems.
Evolution of the Calcium uniporter. Image Credit: Institute for Research in Biomedicine (IRB Barcelona).
This phenomenon occurred around one billion years ago before animals and fungi diverged evolutionarily.
At present, the fungal models being used for analyzing mitochondrial calcium regulation are inadequate, because the existing system is not similar to that of animals. The only fungi that have a system analogous to humans are Chytrids—a divergent class of fungi.
The study was published in the Nature Communications journal.
Numerous processes in cells are controlled by calcium levels. These processes range from division to metabolism. And these levels are, in turn, controlled by calcium transport both into and out of mitochondria, which is the energy hub of the cell.
Hence, the control of calcium levels is a frequent topic of study, and has major implications in the field of biomedicine. As such, fungi are usually the preferred model organisms to analyze this system.
The Comparative Genomics Laboratory of IRB Barcelona, led by Toni Gabaldón, an ICREA researcher, and by the Barcelona Supercomputing Center (BSC-CNS), has demonstrated that the transporter that controls the calcium levels in cells duplicated some one billion years ago, generating a pair of transporter subtypes.
Among these, just one transporter subtype is found in animals, while a majority of the fungi preserve the other one. The aquatic microscopic organisms Chytrids are the only family in the fungal kingdom to have the same transporter subtype as animals.
Hence, if scientists use the calcium transporter of other kinds of fungus as a model, it may result in confused observations and thus wrong conclusions.
Determining how cell systems came about and evolved helps us to understand them and to choose appropriate model organisms for research purposes.”
Toni Gabaldón, ICREA Researcher, Centre for Genomic Regulation, Barcelona Institute of Science and Technology
An extensive computational analysis of more than 1000 organisms
The study employed computational techniques to examine the whole genome of over 1000 organisms, to rebuild the evolution of the calcium transport machinery that occurs in mitochondria. The ancestral origin of this mitochondrial transport system was validated by the analysis. The common ancestor of animals and fungi already had this mitochondrial transport system.
At the Helmholtz Zentrum in Munich (Germany), Fabiana Perochi’s laboratory performed experimental tests that demonstrated that chytrid proteins execute a function similar to that of human beings.
The paradox of fungi that do not transport calcium
Earlier, it had been noted that within the fungi, the calcium transporter serves differently from that present in animals and it also exists in organisms that do not convey calcium to mitochondria. Such an observation was believed to be a paradox.
By reconstructing the evolutionary history of these proteins, we have been able to resolve a paradox and identify the true equivalents of the human system in fungi. This finding opens up new avenues for their use as experimental models.”
Toni Gabaldón, ICREA Researcher, Centre for Genomic Regulation, Barcelona Institute of Science and Technology
Source:
Journal reference:
Pittis, A. A., et al. (2020) Discovery of EMRE in fungi resolves the true evolutionary history of the mitochondrial calcium uniporter. Nature Communications. doi.org/10.1038/s41467-020-17705-4.