Metabolites are essentially organic molecules that are formed or participate during the biochemical reactions that continuously occur in an organism. Over 110,000 metabolites have been identified in the human body.
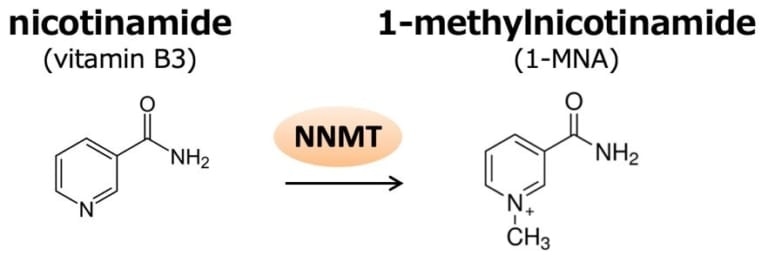
NNMT catalyzes the methylation of nicotinamide to produce 1-methylnicotinamide (1-MNA). 1-MNA can be further oxidized by aldehyde oxidase into, N1-methyl-2- pyridone-5-carboxamide (2py) or N1-methyl-4-pyridone-3-carboxamide (4py), and all three metabolites are excreted in the urine. Upon addition of 1-MNA, the fluorescence intensity of P6A dramatically decreased because of photoinduced electron transfer. P6A also has high selectivity: P6A does not form host–guest complexes with nicotinamide and 2py. Image Credit: Kanazawa University.
Metabolites have a crucial role to play in metabolic syndrome, a situation in which many medical conditions occur at the same time; these conditions include obesity, high blood sugar, and high blood pressure. Metabolic syndrome is linked to a greater risk of developing type-2 diabetes, cardiovascular disease, and many different types of cancers.
The presence of specific metabolites can indicate certain pathological conditions associated with metabolic syndrome. Therefore, efficiently tracking and measuring the presence of metabolites is crucial for early diagnosis.
At present, Tomoki Ogoshi, Atsushi Hirao, and Masaya Ueno (the corresponding authors of the study) from Kanazawa University and collaborators have designed a biosensor for a low-molecular-weight metabolite called 1-MNA. The sensor depends on the physicochemical properties of a channel-like molecule, known as pillar[6]arene.
The team studied 1-MNA (1-methylnicotinamide), a metabolite that was recently found to be present at higher concentrations in aggressive cancer cell lines. Such cancers have boosted the activity of nicotinamide N-methyltransferase (NNMT), where 1-MNA is a by-product. Therefore, identifying 1-MNA could be significant for the timely diagnosis and treatment of these cancers.
Ogoshi and collaborators have proposed that pillar[n]arenes could be utilized as biosensors for metabolites, such as 1-MNA. Pillar[n]arenes are essentially pillar-shaped macrocyclic compounds that have a polygonal cross-section (pentagonal and hexagonal for n = 5 and 6, in that order).
The team discovered that pillar[6]arene (P6A) forms a host-guest complex with the 1-MNAmetabolite; the metabolite can attach to it because the hexagonal cavity within P6A provides the right surroundings for doing so.
The researchers also found that when the 1-MNA metabolite is attached to P6A, the fluorescent reaction of the latter considerably reduces—an impact that can be manipulated as an indicator for the absence or presence of the 1-MNA metabolite (weak or strong fluorescent response, in that order).
Most significantly, the researchers could demonstrate that the P6A fluorescence detection mechanism also works for biological specimens. In particular, they successfully detected the 1-MNA metabolite in urine but with low sensitivity.
According to Ogoshi and collaborators additional experiments “will help to improve the sensitivity and specificity of the biosensors,” and that their study “should contribute to the development of low-cost, easy, and rapid methods for the detection of human metabolites for diagnosis.”
Source:
Journal reference:
Ueno, M., et al. (2020) Pillar[6]arene acts as a biosensor for quantitative detection of a vitamin metabolite in crude biological samples. Communications Chemistry. doi.org/10.1038/s42004-020-00430-w.