Professor Xiang David LI of The University of Hong Kong’s Research Division for Chemistry and Department of Chemistry led a research team to create a novel chemical method for illustrating protein interaction networks in cells.
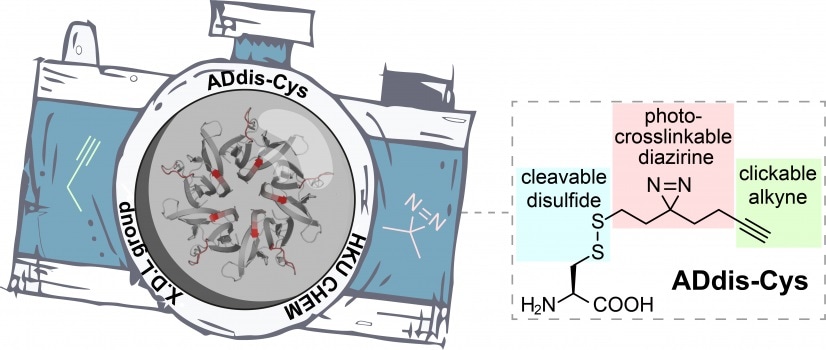
The ADdis-Cys “camera” can simultaneously identify a protein’s interacting partners and pinpoint their binding regions. Image Credit: The University of Hong Kong.
This technique not only helps identify a protein’s interacting partners in a diverse cellular setting but also enables the “visualization” of these protein-protein interactions. The study results were reported recently in the renowned scientific journal Molecular Cell.
Proteins interact with one another in the human body to control almost any biological function, from signal transduction and gene expression to immune response. Thus, dysregulated protein associations usually result in human diseases like Alzheimer’s and cancer.
In modern biology, it is critical to fully comprehend protein interaction networks, as this has implications for disease diagnosis and may aid in the development of therapies.
Two questions must be answered to dissect diverse protein networks: “how” and “who” protein binding occurs. The “who” denotes identifying a protein’s interacting partners, while the “how” denotes the particular “binding regions” that facilitate these interactions.
Addressing this issue is difficult since protein associations are often too transient and unstable to identify. Professor Li’s group previously built a set of tools to “trap” protein-to-protein interactions with a chemical bond to address this problem.
This is viable because these tools have a special light-activated “camera”—a diazirine group that records any binding partner of a protein upon exposure to UV light. After that, the interactions can be analyzed and understood.
Regrettably, the “resolution” of this “camera” was relatively poor, resulting in the loss of critical information on how proteins communicate with one another. For this purpose, Professor Li’s team has now developed a new tool (named ADdis-Cys) with an improved “camera” to boost the “resolution.”
An alkyne handle mounted next to the diazirine allows a “zoom in” to easily see the protein binding regions. ADdis-Cys is the first instrument that can simultaneously detect a protein’s interacting partners and locate its binding regions when combined with cutting-edge mass spectrometry.
As reported in the paper, Professor Li’s lab could thoroughly detect multiple protein interactions—some recognized and some unknown—that are critical for the control of basic cellular processes like gene transcription, DNA damage repair, and DNA replication.
Most significantly, Professor Li’s could identify the binding regions that mediate these protein interactions using ADdis-Cys. This technique could pave the way for the development of chemical modulators that control protein interactions for the treatment of human diseases.
As a research tool, ADdis-Cys may have wide-ranging applications, especially in the diagnosis and treatment of various diseases.
Source:
Journal reference:
Lin, J., et al. (2021) A tri-functional amino acid enables mapping of binding sites for posttranslational-modification-mediated protein-protein interactions. Molecular Cell. doi.org/10.1016/j.molcel.2021.04.001.