UCL scientists have recorded the sharpest images ever of living bacteria, which showed the complex architecture of the protective layer that surrounds most bacteria and makes them more difficult to be destroyed by antibiotics.
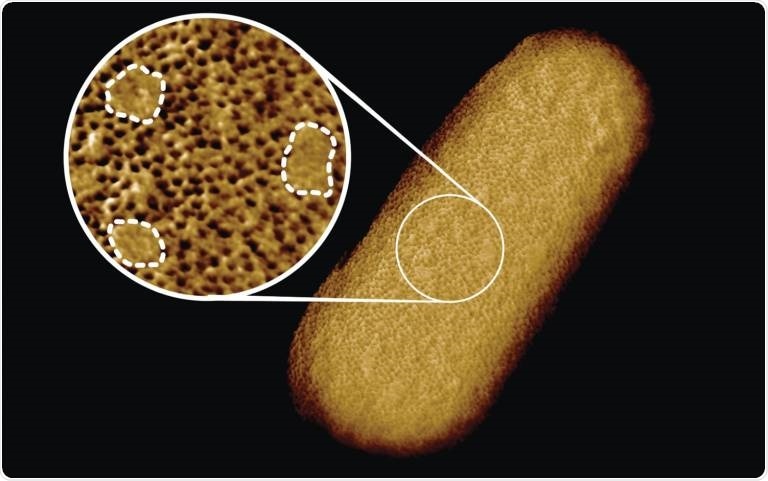
Microscopy image of a living E. coli bacterium, revealing the patchy nature of its protective outer membrane. A densely packed network of proteins is interrupted by smooth, protein-free islands (labeled by dashed lines in the inset). Image Credit: Benn et al. University College London.
The research was a collaborative effort of researchers from the National Physical Laboratory, King’s College London, University of Oxford, and Princeton University. The study demonstrates that bacteria with protective outer layers—known as Gram-negative bacteria—might have stronger and weaker spots on their surface.
The research was published on October 25th, 2021, in the Proceedings of the National Academy of Sciences of the United States of America.
The researchers identified that the protective outer membrane of the bacteria has dense networks of protein building blocks alternated by patches that do not have proteins. But these patches are enriched in molecules with sugary chains (glycolipids) that maintain the outer membrane tight.
This is a vital observation as the tough outer membrane of Gram-negative bacteria stops certain antibiotics and drugs from penetrating the cell. This outer membrane is one of the reasons why antimicrobial resistance of such bacteria (including P. aeruginosa, A. baumannii, and Enterobacteriaceae like E. coli and Salmonella) is considered a greater threat than Gram-positive bacteria like resistant S. aureus (generally known as MRSA).
The outer membrane is a formidable barrier against antibiotics and is an important factor in making infectious bacteria resistant to medical treatment. However, it remains relatively unclear how this barrier is put together, which is why we chose to study it in such detail.”
Bart Hoogenboom, Corresponding Author and Professor, London Centre for Nanotechnology, University College London
Bart Hoogenboom is also a professor at UCL Physics & Astronomy.
By studying live bacteria from the molecular to cellular scale, we can see how membrane proteins form a network that spans the entire surface of the bacteria, leaving small gaps for patches that contain no protein. This suggests that the barrier may not be equally hard to breach or stretch all over the bacterium, but may have stronger and weaker spots that can also be targeted by antibiotics.”
Bart Hoogenboom, Corresponding Author and Professor, London Centre for Nanotechnology, University College London
To comprehend better on this architecture, the researchers ran a tiny needle over living Escherichia coli (E. coli) bacteria, thus “feeling” their overall shape. As the tip of the needle is only a few nanometers wide, it enabled visualization of the molecular structures at the bacterial surface.
The resultant images revealed that the whole outer membrane of the bacteria is packed with microscopic holes developed by proteins that enable the entry of nutrients at the same time hindering toxin entry. Even though it was known that the outer membrane contains numerous proteins, this crowded and immobile nature was unanticipated.
Interestingly, the images also showed numerous patches that did not appear to have proteins. These patches have a glycolipid generally seen on the surface of Gram-negative bacteria along with a different type of pimple-like patch developed when parts of the membrane were flipped inside out owing to mutations.
The presence of these defects is linked with increased sensitivity to bacitracin—an antibiotic generally effective against Gram-positive and not against Gram-negative bacteria.
The textbook picture of the bacterial outer membrane shows proteins distributed over the membrane in a disordered manner, well-mixed with other building blocks of the membrane. Our images demonstrate that that is not the case, but that lipid patches are segregated from protein-rich networks just like oil separating from water, in some cases forming chinks in the armor of the bacteria.”
Georgina Benn, University College London
Georgina Benn carried out microscopy on the bacteria in Professor Hoogenboom’s laboratory. Benn further states, “This new way of looking at the outer membrane means that we can now start exploring if and how such order matters for membrane function, integrity, and resistance to antibiotics.”
The researchers also anticipate that the observations might help elaborate how bacteria can maintain a highly packed, protective barrier permitting rapid growth at the same time. The common bacterium E. coli doubles in size and later divides in 20 minutes under favorable conditions.
The researchers propose that the glycolipid patches might permit for more stretch of the membrane than the protein networks, facilitating the membrane to adapt to the growing size of the bacterium.