Psoriasis is an autoimmune disease that is characterized by patches of red, inflamed skin and painful, scaly rashes. Around 7.5 million Americans suffer from Psoriasis. Even though there exist efficient effective treatments for psoriasis, not every person responds to these therapies—and for most of them, the relief is temporary.
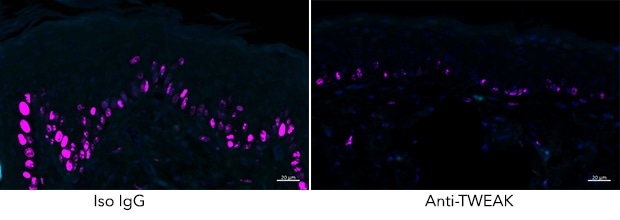
The Croft Lab captured how the proliferation of skin cells, as seen in psoriasis (indicated with purple staining), decreased when TWEAK activity was blocked. Image Credit: Rinkesh Gupta, La Jolla Institute for Immunology.
These therapies don’t reduce disease by 100 percent, and they don’t cure the disease. And if you take patients off those drugs, the disease almost always comes back.”
Michael Croft PhD, Professor, La Jolla Institute for Immunology
Recently Croft and his group from LJI’s Center for Autoimmunity and Inflammation identified a major protein named TWEAK that damages skin cells in psoriasis patients. The results in mice and with human skin cells indicate that by targeting TWEAK psoriasis can be controlled.
We think TWEAK might be considered a potential target for the treatment of psoriasis. It’s good to have this chance to develop a new therapeutic option.”
Rinkesh Gupta PhD, Study First Author and Postdoctoral Fellow, La Jolla Institute for Immunology
The research was published in the Science Immunology journal.
The results were based on Croft Laboratory’s earlier research indicating that TWEAK can interact with keratinocyte—the common type of skin cell. By analyzing TWEAK-deficient mice, the scientists identified that TWEAK is an inducer of inflammation in a psoriasis model.
The current research demonstrates that TWEAK does not work alone. By examining, human keratinocytes, scientists found that TWEAK combines two other proteins—tumor necrosis factor (TNF) and interleukin-17 (IL-17)—to induce inflammation. These proteins regulate the production of inflammatory molecules and the expression of more inflammation-linked proteins in psoriasis patients.
The fact that they work together suggests the disease is essentially driven by all three of those particular proteins at the same time. The primary implication is that TWEAK will also be a good drug target. as has already been proven for TNF and IL-17.”
Michael Croft PhD, Professor, La Jolla Institute for Immunology
To analyze this, the scientists utilized a mouse model of psoriasis to compare how a TWEAK-inhibitor aligned up to therapies inhibiting IL-17 or TNF.
The observations indicate that the group is on the right path. “If you inhibit TWEAK from working on its receptor on keratinocytes, you get the same therapeutic effect as when you inhibit TNF or IL-17,” details Gupta. This observation is particularly encouraging as IL-17 and TNF are both FDA-approved drug targets for psoriasis.
Even though human clinical trials need to be carried out, Croft foresees a future for TWEAK inhibitors as therapies for numerous skin diseases. Croft also states, “We think TWEAK is involved in skin inflammation in general.”
Croft’s laboratory is currently analyzing the role of TWEAK in atopic dermatitis—also known as eczema, a very common kind of skin inflammation, particularly in babies and young children. Croft states that while psoriasis and eczema are separate diseases, they have certain similarities and there do not exist better treatments for eczema.
“There’s certainly a lot of room for improvement in the treatment of atopic dermatitis patients,” concluded Croft.
Source:
Journal reference:
Gupta, R. K., et al. TWEAK functions with TNF and IL-17 on keratinocytes and is a potential target for psoriasis therapy. Science Immunology. doi.org/10.1126/sciimmunol.abi8823.