Acute myeloid leukemia, a type of cancer that affects blood and bone marrow cells, is extremely hazardous. Patients under the age of 60 make up more than half of those who die. For patients above the age of 60, this percentage jumps to 85%.
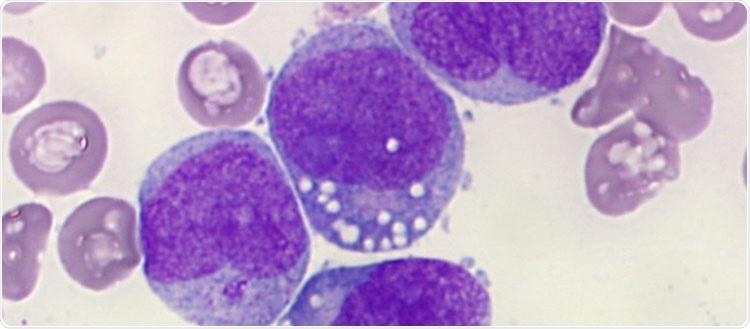
Selective activation of the AMPK enzyme would lead to the death of leukemia cells (in purple in this image). Image Credit: © Jérôme Tamburini/UNIGE.
Furthermore, in an animal model of the condition, the researchers have successfully utilized this energy gap: a combination of two medications, one of which is currently on the market, has shown promise.
However, their efficacy on leukemia stem cells, which can evade numerous treatments and re-grow tumors, has yet to be determined. The findings were published in the journal Cell Reports.
 Image Credit: Kateryna Kon/Shutterstock.com
Image Credit: Kateryna Kon/Shutterstock.com
Jérôme Tamburini, an associate professor in the Department of Medicine and the Translational Research Centre in Onco-Haematology (CRTOH) of the UNIGE Faculty of Medicine, as well as a professor at the Swiss Cancer Center Léman (SCCL) and Université de Paris, studies the energetic frameworks of tumor cells in acute myeloid leukemia. Tamburini is particularly interested in the AMPK cell signaling system.
AMPK is the main detector of the cells energy level. This pathway is activated when energy is lacking and initiates the degradation of certain nutrients to produce the necessary energy – a process called catabolism. As without energy, no cell can survive, could it be possible to selectively manipulate this mechanism in tumor cells to cause their destruction, while preserving healthy cells?”
Jérôme Tamburini, Associate Professor, Translational Research Centre in Onco-Haematology, Faculty of Medicine, University of Geneva
In 2015, Jérôme Tamburini and his colleagues at Inserm in Paris collaborated with the GlaxoSmithKline (GSK) laboratory on the creation of GSK621, a pharmaceutical component that appeared to be an effective AMPK activator in vitro.
After this initial proof of principle, we had to decipher the biochemical mechanisms at work in order to understand them in detail, and in particular which cellular pathways did GSK621 activate in leukemia cells, the first step in hoping to exploit this phenomenon for therapeutic purposes.”
Jérôme Tamburini, Associate Professor, Translational Research Centre in Onco-Haematology, Faculty of Medicine, University of Geneva
An effective combination of two drugs
The initial step was to analyze gene expression in human tumor cells, which revealed that an enzyme called PERK was highly active in the presence of GSK621. This is an important component of the endoplasmic reticulum’s stress response, which is an intracellular structure that specializes in protein and lipid metabolism.
The activation of AMPK thus triggers the activation of PERK, followed by a chain of reactions leading to apoptosis, the programmed death of the cell. In addition, the activation of AMPK by GSK621 sensitizes the cells to the effects of another pharmacological drug, the venetoclax, which is now widely used to treat acute myeloid leukemia, although with limited effectiveness when used alone.”
Jérôme Tamburini, Associate Professor, Translational Research Centre in Onco-Haematology, Faculty of Medicine, University of Geneva
The researchers subsequently combined the two medications in mice carrying human tumor cells and discovered that the mixture was substantially more powerful than monotherapy in controlling tumor growth. While GSK621 was not intended to be a medicine, other compounds that stimulate the AMPK pathway are currently being evaluated in clinical trials to treat metabolic illnesses.
Tamburini also explains,
Understanding the mechanism involved has brought to light potential therapeutic targets that were previously unknown. “We will now be able to review all the drugs known to have an effect on these pathways and determine which combinations would be the most effective.”
What about leukemic stem cells?
Leukemic stem cells are a small number of cells within a tumor that can only be identified by their potential to re-spread the tumor after effective treatment. These cells are the primary cause of recurrence and are resistant to only a handful of the standard leukemia treatments.
Furthermore, there is presently little evidence to establish the impact of substantial AMPK activation on them.
The authors conclude by stating,
Before testing drug combinations targeting this AMPK/PERK mechanism in human beings, we need to determine their effect on leukemic stem cells.”
Source:
Journal reference:
- Grenier, A., et al. (2022) AMPK-PERK axis represses oxidative metabolism and enhances apoptotic priming of mitochondria in acute myeloid leukemia. Cell Reports. doi.org/10.1016/j.celrep.2021.110197.