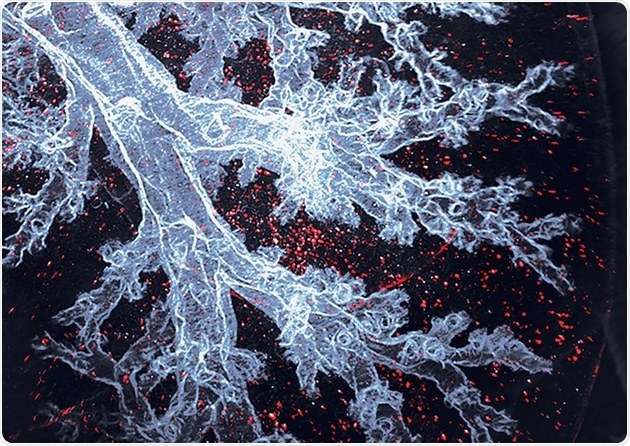
Alveolar macrophages are the immune cells that specifically live in the lung air sacs, here visible as red dots in an otherwise transparent mouse lung. Image Credit: © Sara Gholamhosseinian Najjar and Michaela Burkon.
Macrophages are immune cells that are important for tissue repair, immune response, and the elimination of cancer cells. Researchers consider macrophages as likely living therapeutics. But macrophages need to be multiplied to more numbers in lab culture without losing their unique purposes so that they can be efficiently used for therapies. Until now, it was uncertain whether this is even possible.
Now, a group of researchers from Dresden and Marseille reports that macrophages cultured for long durations in lab conditions could work normally when transported back into the body. They are also indistinguishable from the cells that did not leave the tissue ever. The results lead the path to new macrophage-based cell treatments. The research was published on February 24th, 2022, in the journal Nature Immunology.
Macrophages—immune cells—exist in all organs of the body. They serve as the guardians of tissue, fostering other cells and eliminating detrimental substances like bacteria, cellular debris, and even tumor cells. Hence, macrophages have the interest of researchers as likely new living drugs to fight infections, cure damaged organs, and battle against cancer.
To achieve this, however, cells need to be grown, in large numbers, outside of the body. Until now, this has been challenging for macrophages. Above all, there were strong speculations that lab conditions may make them lose their unique capacities.
Cell culture, which is a process of multiplying cells in the laboratory, is a common procedure that allowed great progress in medicine and biology over the years. However, cells cultured in the lab are removed from their natural surroundings and the physical signals that seem necessary for them to function.
Cells are cultured on plastic culture dishes, and they are cleansed in artificial nutrient solutions. They need to adjust to these new environments, which is a real culture shock.
We wanted to know exactly how the cells change in prolonged cell culture and whether these changes are permanent or not.”
Michael Sieweke, Humboldt Professor, Technische Universität Dresden
The cell culture shock
Prof. Sieweke’s team at the Center for Regenerative Therapies Dresden (CRTD) at TU Dresden and the Center of Immunology of Marseille Luminy (CNRS, INSERM, Aix-Marseille University) analyzed mouse lung macrophages—the immune cells that live in the air sacs of the lung naturally.
Researchers managed to culture the cells under laboratory conditions to large numbers over several months. Although their appearance and general features were not affected when observed more closely, it became evident that the cells had actually obtained many changes to adjust to the new environment.
Every cell in our body has the same set of genes, but the cells differ in which genes are turned on and which are kept off. One can think of it as the molecular fingerprint of the cell – a unique combination of turned-on genes that distinguish, e.g., a lung macrophage from an intestine macrophage and a brain cell.”
Sethuraman Subramanian, Study Author, Technische Universität Dresden
The team has associated the gene pattern in the cells grown in the lab with their counterparts from the lung. They have noticed considerable differences.
“This was to be expected. Living on a plastic surface and having all the nutrients readily available is quite different from natural conditions. The cells had to get used to it and did so by changing the status of more than 3,000 genes. The question that truly interested us was whether these changes can be reversed,” explains Prof. Sieweke.
Forgetting the culture
The team transported the macrophages grown in the lab back into their natural environment, which is the mouse lungs. Elaborated comparisons revealed that the cells cultured in the lab were indistinguishable from their counterparts did not leave the lungs ever.
We were surprised to see that the substantial adaptations that the macrophages made to live in the laboratory have proven to be completely reversible. The lab-cultured macrophages had forgotten about the time they spent in the lab and fully assumed their normal function and status in the lung, oblivious of their previous culture shock.”
Clara Busch, Study Author, Technische Universität Dresden
Cell therapies of the future
Even though the study was performed in mice, it has very hopeful effects for human treatments. The capacity to transport the macrophages from the cell culture to their natural environment indicates promising potential for future macrophage-based cell therapies.
The lung macrophages can be multiplied in the lab conditions and tailored experimentally for combatting a particular disease before delivering to the patient’s lungs where they can instantly start performing their function. This setup can be used to cure fibrotic disease, cancer, or diseases like COVID-19 in the lung and consequently in other organs as well.
“This study started long before the beginning of the pandemic but demonstrates again that fundamental research can serve as a source of future therapeutic applications,” Prof. Sieweke concludes.
Source:
Journal reference:
Subramanian, S., et al. (2022) Long-term culture-expanded alveolar macrophages restore their full epigenetic identity after transfer in vivo. Nature Immunology. doi.org/10.1038/s41590-022-01146-w.