A recent study published in Nature Communications by an international research group suggests that people infected with COVID-19 may have several distinct SARS-CoV-2 variants stashed away from the immune system in various parts of the body. According to the authors of the study, this may make total virus clearance from an infected person’s body, whether by their antibodies or therapeutic antibody therapies, even more difficult.
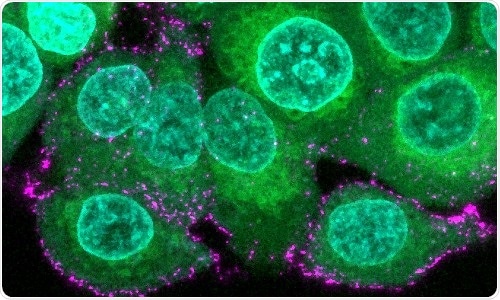
Image of human epithelial cells (green with blue nuclei) are incubated with synthetic SARS-CoV-2 virions (magenta) to study infection and immune evasion. Image Credit: Oskar Staufer and Max Planck Institute for Medical Research, Germany.
COVID-19 is still sweeping the globe, causing hospitalizations and deaths while wreaking havoc on communities and economies around the world. The original virus from Wuhan was replaced by consecutive variants of concern (VoC), which increasingly evaded immune protection provided by vaccination or antibody treatment options.
An international group led by Professor Imre Berger at the University of Bristol and Professor Joachim Spatz at the Max Planck Institute for Medical Research in Heidelberg, both Directors of the Max Planck Bristol Centre of Minimal Biology, demonstrate how the virus can develop distinctively in various cell types and modify its immunity in the very same infected host. The two new studies were published in the journal Nature Communications.
The researchers wanted to look into the role of a custom-made pocket in the SARS-CoV-2 spike protein in the virus’s infection cycle. The pocket, discovered by the Bristol team in a previous discovery, was critical in viral infectiousness.
An incessant series of variants have completely replaced the original virus by now, with Omicron and Omicron 2 dominating worldwide. We analyzed an early variant discovered in Bristol, BrisDelta. It had changed its shape from the original virus, but the pocket we had discovered was there, unaltered.”
Imre Berger, Professor, University of Bristol
BrisDelta, which appears as a small subpopulation in clinical samples, has seemed to infect specific cell-types stronger than the virus that influenced the first wave of infections.
Dr Kapil Gupta, who is a lead author of the BrisDelta study, elaborates, “Our results showed that one can have several different virus variants in one’s body. Some of these variants may use kidney or spleen cells as their niche to hide, while the body is busy defending against the dominant virus type. This could make it difficult for the infected patients to get rid of SARS-CoV-2 entirely.”
To decipher viral mechanisms at work, the team used cutting-edge synthetic biology methods, cutting-edge imaging, and cloud computing. To better understand the mechanism of the pocket, the researchers created synthetic SARS-CoV-2 virions in a test tube that are virus replicates but have a strong benefit in that they are safe since they do not multiply in human cells.
Researchers were able to examine the actual mechanism of the pocket in viral infection using these artificial virions. They demonstrated that when a fatty acid was bound, the spike protein that adorned the virions changed shape. This “shape” switching method effectively hides the virus from the immune system.
Dr Oskar Staufer, lead author of this study explains, “By ‘ducking down’ of the spike protein upon binding of inflammatory fatty acids, the virus becomes less visible to the immune system. This could be a mechanism to avoid detection by the host and a strong immune response for a longer period of time and increase total infection efficiency.” Staufer was also a joint member of the Max Planck Institute in Heidelberg and the Max Planck Centre in Bristol.
It appears that this pocket, specifically built to recognize these fatty acids, gives SARS-CoV-2 an advantage inside the body of infected people, allowing it to multiply so fast. This could explain why it is there, in all variants, including Omicron.”
Imre Berger, Professor, University of Bristol
“Intriguingly, the same feature also provides us with a unique opportunity to defeat the virus, exactly because it is so conserved–with a tailor made antiviral molecule that blocks the pocket,” added Berger.
Halo Therapeutics, the latest University of Bristol spin-off founded by the authors, is pursuing a similar strategy to create pocket-binding pan-coronavirus antivirals.
Source:
Journal reference:
Gupta, K., et al. (2022) Structural insights in cell-type specific evolution of intra-host diversity by SARS-CoV-2. Nature Communications. doi.org/10.1038/s41467-021-27881-6.