A recent study shows that compressing cancer cells through tight spaces makes them more violent and helps them avoid cell death. The study was published in eLife.
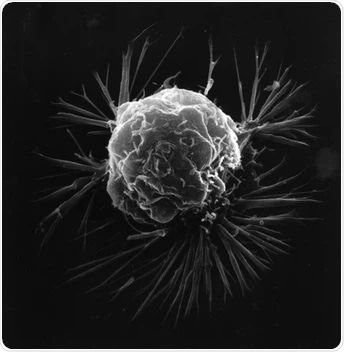
A breast cancer cell, photographed using a scanning electron microscope to produce a 3-dimensional image. Image credit: National Cancer Institute (public domain)
The results present how cancer cells are more possible to spread or metastasize, under mechanical stress. Although metastasis is the reason for a majority of cancer deaths, there are no cures available currently. The new findings, however, might aid scientists in developing new methodologies to cure or avoid metastasis.
It could be a tight compression for cancer cells to escape their tumor or get passage into tiny blood vessels, known as capillaries, eventually to spread across the body. The cells need to collapse and alter their form to get this process done, in an approach known as confined migration. As cancer cells spread, they must also prevent identification and damage by the immune system.
Mechanical stress can cause cancer cell mutations, as well as an uncontrolled increase in cell numbers and greater tissue invasion. We wanted to know if the mechanical stress of confined migration makes cancer cells more likely to metastasize, and how this happens.”
Deborah Fanfone, Study First Author and Postdoctoral Fellow, Cancer Research Center of Lyon
To get a solution, Fanfone and co-workers enforced cancer cells of the human breast through a membrane that has tiny, three-micrometer-sized holes to trigger a confined migration environment.
After only one passage via the membrane, the researchers discovered that the cells got more mobile and immune to anoikis—a form of programmed cell death that happens when cells get detached from the neighboring network of proteins and other molecules that back them (the extracellular matrix). The cells were also capable of escaping destruction through immune natural killer cells.
Additional experiments revealed that increased expression of inhibitory-of-apoptosis proteins (IAPs) increased the cancer cells’ resistance to anoikis. Curing the cancer cells with SMAC mimetic—a new type of cancer drug, which degrades IAPs—eliminated this protection.
Then, the researchers studied how breast cancer cells, which had gone through confined migration, behave while administered to immune-suppressed mice. They discovered that these mice developed increased lung metastases than mice that were administered with breast cancer cells, which have not been exposed to confined migration.
By mimicking confined migration, we’ve been able to explore its multifaceted effects on cancer aggressiveness. We’ve shown how the process boosts survival in cancer cells and makes them more prone to forming deadly metastases.”
Gabriel Ichim, Study Senior Author and Leader, Cancer Cell Death Team, Cancer Research Center of Lyon
The authors state that these findings might result in extra studies of potential metastasis treatments, like treatments that soften tumors to decrease mechanical stress upon cancer cells, or that block IAPs. They comprise SMAC mimetics that are now being assessed in clinical trials as a potential new treatment method.
Source:
Journal reference:
Fanfone, D., et al. (2022) Confined migration promotes cancer metastasis through resistance to anoikis and increased invasiveness. eLife. doi.org/10.7554/eLife.73150.