RIKEN scientists have extracted and grown stem cells that give way to the mouse yolk sac for the first time in the laboratory, boosting the potential of generating mouse embryos from stem cells in the coming years.
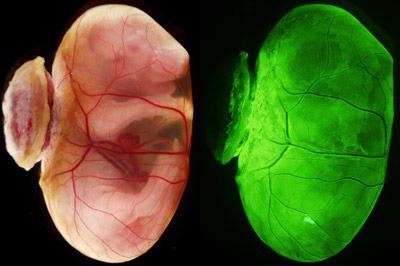 Figure 1: Bright-field microscopy image (left) and fluorescence microscopy image (right) of an E18.5 embryo with a yolk sac. When blastocysts that were depleted in primitive endoderm cells and injected with primitive endoderm stem cells were implanted in the uteri, they developed living offspring with yolk sacs (structure encasing the embryo), just like wild-type blastocysts. From Ref. 1. Reprinted with permission from AAAS. Image Credit: RIKEN
Figure 1: Bright-field microscopy image (left) and fluorescence microscopy image (right) of an E18.5 embryo with a yolk sac. When blastocysts that were depleted in primitive endoderm cells and injected with primitive endoderm stem cells were implanted in the uteri, they developed living offspring with yolk sacs (structure encasing the embryo), just like wild-type blastocysts. From Ref. 1. Reprinted with permission from AAAS. Image Credit: RIKEN
After uniting with sperm, an egg grows into a blastocyst, which is an early embryo. It is made up of only three types of cells: epiblast cells (which form the embryo), trophoblast cells (which produce the placenta), and primitive endoderm cells (producing major parts of the yolk sac).
Although epiblast and trophoblast stem cells have been identified, no one has yet succeeded in generating stem cells for primitive endoderm cells.
Yasuhide Ohinata and colleagues from the RIKEN Center for Integrative Medical Sciences have now created primitive endoderm stem cells in mice, which form the yolk sac that feeds the growing blastocyst until the placenta takes control.
Researchers did this by putting mouse blastocysts in different cultures and figuring out what circumstances allowed primitive endoderm stem cells to thrive. They used recombinant proteins and tiny chemicals to disrupt important functioning signaling pathways in blastocysts, and then examined the resultant cells.
These cells properly complement the embryonic development of primitive-endoderm-cell-depleted blastocysts in chimeras, according to the researchers. After being transferred into the uteri, these blastocysts matured into normal offspring.
This was not a simple demonstration in live mice.
There’s a big leap between in vitro culture and in vivo function, as it’s impossible to know whether established stem cells will function in vivo without transplanting them. We were fortunate in that we were able to achieve the expected results in vivo.”
Yasuhide Ohinata, Center for Integrative Medical Sciences, RIKEN
The researchers want to generate embryos artificially in the future. “Our ultimate goal is to reconstitute embryos that can develop from stem cells alone,” says Ohinata. “Primitive endoderm stem cells are essential for this, which is why we devoted so much effort to establishing a generally overlooked stem-cell type.”
The scientists used a combination of the three stem cell types to create embryo-like structures in vitro. When these were deposited in uteri, they produced descendants with yolk-sac-like structures, but they did not develop into normal embryos.
While these results represent a major step forward in artificial embryo reconstitution, we will continue to pursue research to mimic embryos more precisely using stem cells.”
Yasuhide Ohinata, Center for Integrative Medical Sciences, RIKEN
Due to the variations in embryonic development in mice and humans, Ohinata emphasizes that this accomplishment may not be readily relevant to people, however, he adds, “To understand human and mouse development in an integrated manner, we’re conducting research using pigs, which are thought to retain human-type developmental mechanisms.”
Source:
Journal reference:
Ohinata, Y., et al. (2022) Establishment of mouse stem cells that can recapitulate the developmental potential of primitive endoderm. Science. doi.org/10.1126/science.aay3325.