Reviewed by Danielle Ellis, B.Sc.May 25 2022
According to a new study published May 24th, 2022, in the open-access journal PLOS Biology by Alena Sumová and coworkers of the Czech Academy of Sciences in Prague, genes within the fetus’s developing clock respond to rhythmic behavior in the mother during fetal development, first before the biological clock starts ticking on its own. The results add to the knowledge of how the internal clock develops and might have significance for the treatment of premature newborns.
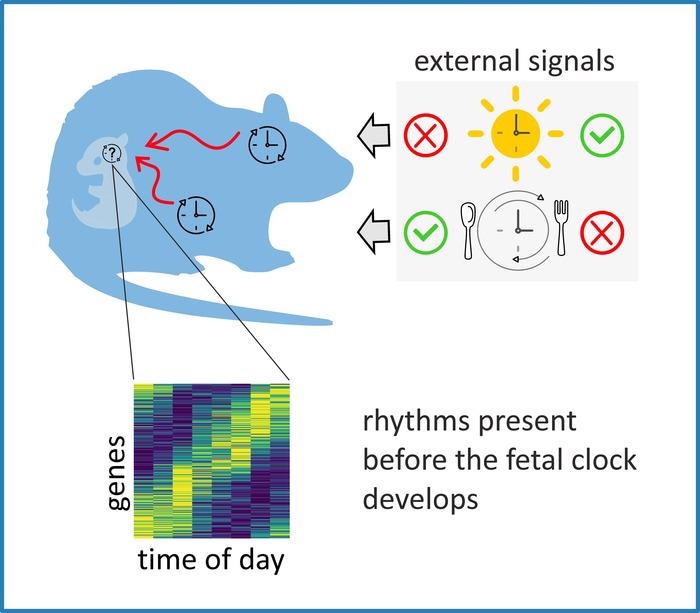 Maternal signals drive rhythmicity in the fetal suprachiasmatic nuclei before their molecular clock develops. Image Credit: Alena Sumová and Martin Sládek (CC-BY 4.0, https://creativecommons.org/licenses/by/4.0/).
Maternal signals drive rhythmicity in the fetal suprachiasmatic nuclei before their molecular clock develops. Image Credit: Alena Sumová and Martin Sládek (CC-BY 4.0, https://creativecommons.org/licenses/by/4.0/).
The suprachiasmatic nuclei (SCN), which are located in the hypothalamus, are the body’s master timekeepers. The activity of many other genes, both locally and elsewhere in the body, is governed by the rhythmic activity of genes in SCN cells, impacting a wide range of circadian rhythmic behavior, including eating and sleeping.
However, rhythmic gene activity begins late in fetal development, raising questions of whether maternal factors entrain gene activity in the SCN before birth.
To find out, the researchers studied the pattern of gene activity in SCN tissue from growing fetuses in pregnant rats housed in the dark in two different settings. The SCNs of control rats were undamaged, and they had full access to food, whereas the SCNs of lesioned rats were disturbed, but their food availability was limited to eight hours per day to impose a circadian rhythm on their activity that their SCNs could not support.

Image Credit: cooperr/Shutterstock
Researchers discovered that among both sets of fetuses’ SCNs, there was a small number of genes whose timing patterns varied and a considerably greater number of genes whose activity oscillated in lockstep. Many of the later genes are associated with two primary processes: neural formation and neuronal function, possibly reflecting the SCN’s continuing development as it lines itself up for a mature function in the one case, and the earliest expression of that function in the second situation.
Our data reveal that in development in the fetal suprachiasmatic nuclei, maternal stimuli may substitute for an absent inter-cellular web of synapses and drive cell-population rhythms before the SCN clock fully matures.”
Alena Sumová, Laboratory of Biological Rhythms, Institute of Physiology of the Czech Academy of Sciences, Prague
These findings may have consequences for premature human newborns, Sumová continued, because the rats used in the study had a gestational period of roughly 21 days and the fetuses were assessed at 19 days.
The unexpected broadness and specificity of responsiveness of the SCN cells to maternal signals stresses the importance of a healthy maternal circadian system during pregnancy, and points at the potential impact of the absence of such signals in prematurely delivered children.”
Alena Sumová, Laboratory of Biological Rhythms, Institute of Physiology of the Czech Academy of Sciences, Prague
“Our study reveals that distinct maternal signals rhythmically control a variety of neuronal processes in the fetal rat suprachiasmatic nuclei before they begin to operate as the central circadian clock. The results indicate the importance of a well-functioning maternal biological clock in providing rhythmic environment during the fetal brain development,” Sumová concludes.
Source:
Journal reference:
Greiner, P., et al. (2022) Early rhythmicity in the fetal suprachiasmatic nuclei in response to maternal signals detected by omics approach. PLOS Biology. doi.org/10.1371/journal.pbio.3001637.