Image Credit: David Song/NYU
The studies showed how a group of novel molecules influenced by the human immune system inactivates numerous viruses, including Zika and chikungunya, in a new study published on August 2nd, 2023, in the journal ACS Infectious Diseases. Their approach may not only result in drugs that can be used against a wide range of viruses, but it may also aid in the fight against antiviral resistance.
The Urgent Need for New Antivirals
Viruses have a variety of proteins on their surfaces that are frequently targeted by therapeutics such as monoclonal antibodies and vaccines. However, targeting these proteins has limitations because viruses evolve quickly, altering the properties of the proteins and making treatments less effective.
These limitations were highlighted when new SARS-CoV-2 variants surfaced that were resistant to both drugs and vaccines developed to combat the original virus.
There is an urgent need for antiviral agents that act in new ways to inactivate viruses. Ideally, new antivirals won’t be specific to one virus or protein, so they will be ready to treat new viruses that emerge without delay and will be able to overcome the development of resistance. We need to develop this next generation of drugs now and have them on the shelves in order to be ready for the next pandemic threat—and there will be another one, for sure.
Kent Kirshenbaum, Study Senior Author and Professor, Chemistry, New York University
Drawing Inspiration From Human Immune Systems
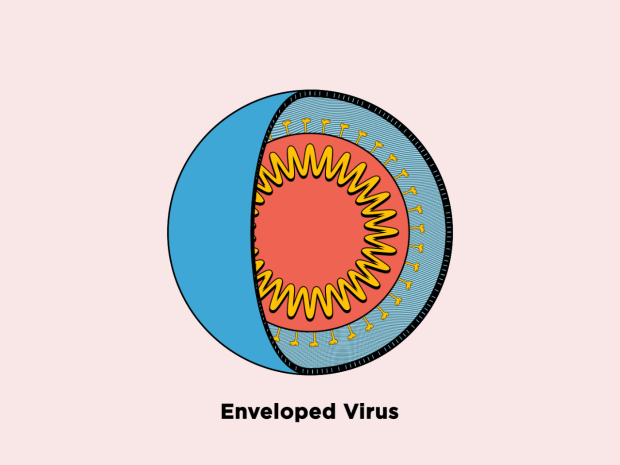
The body’s first line of defense against bacteria, fungi, and viruses is the production of antimicrobial peptides by the innate immune system in the fight against pathogens. Most pathogenic viruses are enclosed in lipid membranes, and antimicrobial peptides function by rupturing or bursting these membranes.
Antimicrobial peptides can be created in the lab, but because they degrade quickly and can be harmful to healthy cells, they are rarely used to treat infectious diseases in humans. Instead, scientists have created synthetic substances called peptoids that resemble peptides chemically but are better able to penetrate viral membranes and are less likely to degrade.
We began to think about how to mimic natural peptides and create molecules with many of the same structural and functional features as peptides, but are composed of something that our bodies won't be able to rapidly degrade.”
Kent Kirshenbaum, Study Senior Author and Professor, Chemistry, New York University
The researchers looked into seven peptoids, many of which were discovered in the lab of Stanford co-author Annelise Barron. The NYU researchers investigated the antiviral effects of peptoids on four viruses: three with membranes (Rift Valley fever, Zika, and chikungunya) and one without (coxsackievirus B3).
“We were particularly interested in studying these viruses as they have no available treatment options,” notes Patrick Tate, a chemistry Ph.D. Student at NYU and the study’s first author.
How Peptoids Disrupt Viral Membranes and Avoid Other Cells
Because lipids are obtained from the host to form membranes, the membranes surrounding viruses are made of different molecules than the viruses themselves. Phosphatidylserine, for example, is found in the membrane of viruses but is sequestered towards the interior of human cells under normal conditions.
“Because phosphatidylserine is found on the exterior of viruses, it can be a specific target for peptoids to recognize viruses, but not recognize—and therefore spare—our own cells. Moreover, because viruses acquire lipids from the host rather than encoding from their own genomes, they have better potential to avoid antiviral resistance,” noted Tate.
The investigators tested seven peptoids against the four viruses. They discovered that peptoids inactivated all three enveloped viruses—Rift Valley fever, Zika, and chikungunya—by obstructing the virus membrane, but not coxsackievirus B3, the only virus that does not have a membrane.
Additionally, the peptoids were more effective against the chikungunya virus, which had a membrane with a higher concentration of phosphatidylserine. The peptoids did not disrupt a membrane made entirely of a different lipid called phosphatidylcholine, indicating that phosphatidylserine is essential for peptoids to reduce viral activity.
“We’re now starting to understand how peptoids actually exert their antiviral effect—specifically, through the recognition of phosphatidylserine,” Tate adds.
The investigators are continuing pre-clinical studies to assess the possibility of these molecules in combating viruses and to determine whether they can overcome resistance development. Their peptoid-focused strategy may hold great promise for treating a wide range of viruses with difficult-to-treat membranes, such as SARS-CoV-2, Ebola, and herpes.
Enveloped Virus Membrane Disruption
Video Credit: David Song/NYU
Source:
Journal reference:
Tate, P. M., et al. (2023) Peptidomimetic Oligomers Targeting Membrane Phosphatidylserine Exhibit Broad Antiviral Activity. ACS Infectious Diseases. doi.org/10.1021/acsinfecdis.3c00063.