I am Michael Santos, Senior Vice President at the Foundation for the National Institutes of Health and Director of the GeneConvene Global Collaborative. GeneConvene advances informed decision-making about genetic biocontrol approaches for public health.
GeneConvene is a continuation of almost 20 years of work at FNIH supporting new approaches to preventing mosquito-borne diseases like malaria and dengue. I joined the team four years ago from the Bill & Melinda Gates Foundation, where I was involved in funding research in this area.
What is World Mosquito Day, and why was it created?
World Mosquito Day commemorates experiments showing that blood-feeding mosquitoes transmit malaria. This fundamental understanding informs the many approaches to protect people from the bites of infectious mosquitoes, including genetic biocontrol approaches.
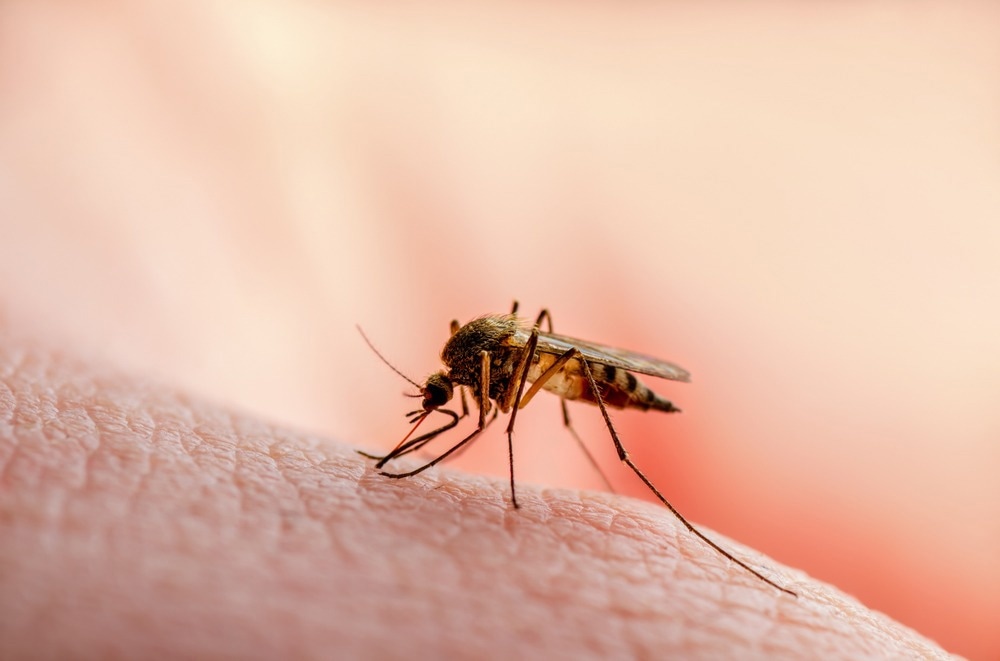
Image Credit: nechaevkon/Shutterstock.com
Malaria is an infectious disease that continues to persist worldwide. Why is malaria so difficult to eradicate?
The parasites that cause malaria and mosquitoes that can transmit those parasites are widespread across the globe, with billions of people at risk.
There has been some progress against malaria: deaths due to malaria declined from 897,000 in 2000 to 577,000 in 2015. This progress was primarily due to expanded access to insecticide-treated bed nets, indoor insecticide spraying, and access to malaria diagnosis and treatment. Increasing access required funding – global investment in malaria control increased from about $500 million in 2000 to more than $3 billion by 2010.
Unfortunately, malaria deaths stayed relatively constant after 2015 until increasing in 2020, likely due to the effects of the SARS-CoV-2 (COVID-19) pandemic. Funding has also been roughly constant since 2010. This stalling of progress is one of the key motivations for the development of new tools to control and eliminate malaria, especially tools that protect more people for the same investment.
One solution that has been raised is to deploy genetically engineered mosquitoes; could you explain how this would work?
Researchers have shown that they can genetically engineer mosquitoes in ways that could reduce the transmission of disease. One approach is to engineer the mosquitoes to reduce the overall number of mosquitoes so that people get bitten less frequently. Another approach is to engineer the mosquitoes so that they can’t transmit the disease; it’s even possible to combine these approaches.
After mosquitoes are genetically engineered, they could be bred and released into the environment, where they would mate with wild mosquitoes. In some approaches, repeated releases would be required to maintain the effect. In other approaches, called gene drive, the mosquitoes would be engineered to pass on the genes in a way that the effect could persist even after releases stop.
There have already been studies of genetically modified mosquitoes in some countries, and they have been approved by the government in Brazil for use there. None of those studies has been of gene drive, only the kinds of approaches that require repeated releases.
Are there any foreseeable problems with this technology?
An important part of government decisions about genetically engineered mosquitoes is a process called risk assessment. In that process, potential problems are identified, and the likelihood of those risks are estimated. In many cases it is possible for researchers to assess the potential risks experimentally, and in other cases risks are assessed based on experience and modeling.
Because the risks are specific to the approach and location, risk assessment is done for every case individually rather than for technology classes generically.
Why do you think a fear remains surrounding genetically engineered organisms?
Surveys show a spectrum of views about genetic engineering, depending on many factors, such as the group surveyed and the engineering application. There is a growing body of research specifically on views about the potential use of genetic biocontrol of mosquitoes to reduce malaria transmission, which typically shows concerns about potential risks and interest in potential benefits. It is important for scientists and government officials to consider these perspectives in their approaches.
What efforts are the GeneConvene Global Collaborative making in this space?
GeneConvene helps identify and address key questions, provides technical advice, strengthens capacity, and shares information. We work with partners to develop guidance, such as the World Health Organization’s Guidance Framework for Testing of Genetically Modified Mosquitoes, offer training on biological and regulatory science, and provide a freely accessible resource with scientific research, media coverage, and answers to common questions at the GeneConvene Virtual Institute.
Many countries already have experience regulating genetically modified organisms and biocontrol interventions. There are also global frameworks like the UN’s Convention on Biological Diversity, which includes a treaty about biosafety regulation. In addition, the African Union supports member states to build their capacity with assistance from the African Union Development Agency–NEPAD. GeneConvene helps build on these foundations to support informed decision-making by all stakeholders.

Image Credit: Justinboat.29/Shutterstock.com
In your opinion, what other efforts should be taken to improve communications around the biotechnology-based solutions to tackle malaria, and, more broadly, infectious disease?
First and foremost, I hope we can raise awareness about the impact of malaria: robbing half a million families every year of the lives of their children.
There are opportunities right now to increase access to life-saving interventions, like bed nets and malaria treatments, by increasing funding to health systems and malaria control programs.
It’s also important to recognize that innovation is required to accelerate progress. Already, mosquitoes and the parasites that cause malaria are developing resistance to our existing tools. New insecticides, diagnostics, and drugs are being developed in response, but they are often more expensive. There has been exciting progress recently with the first generation of malaria vaccines, but these also have limitations. New approaches that can complement the existing tools – especially low-cost, easy-to-use new approaches – are needed to save more lives sooner.
Scientific research has the potential to give people new options to protect themselves and their families from infectious diseases like malaria. It is up to governments, communities, and individuals to decide how best to use those options.
What are the next steps for the GeneConvene Global Collaborative? Where can our readers go to stay up to date with your efforts?
GeneConvene continues our work to support informed decision-making, and I would encourage anyone who wants to learn more to visit the GeneConvene Virtual Institute, which is continuously updated with the latest information and provides the opportunity to subscribe to our weekly newsletter highlighting research, media coverage, and events.
Please provide links to any materials that may be relevant to our audience.
1. The GeneConvene program website, which describes GeneConvene itself: www.geneconvene.org
2. The GeneConvene Virtual Institute website, which provides information on genetic biocontrol: www.geneconvenevi.org
About Dr. Michael Santos
 As Senior Vice President and Chief Population Health Science Officer for the Foundation for the National Institutes of Health (FNIH), Dr. Santos’ work focuses on advancing biomedical innovation to improve health and health equity globally. He is the Director of the GeneConvene Global Collaborative, an FNIH initiative supporting informed decision-making about genetic biocontrol approaches for public health.
As Senior Vice President and Chief Population Health Science Officer for the Foundation for the National Institutes of Health (FNIH), Dr. Santos’ work focuses on advancing biomedical innovation to improve health and health equity globally. He is the Director of the GeneConvene Global Collaborative, an FNIH initiative supporting informed decision-making about genetic biocontrol approaches for public health.
Before joining the FNIH, at the Bill & Melinda Gates Foundation he developed investment strategies to accelerate R&D addressing the biggest drivers of health inequity, including malaria, tuberculosis, HIV, and maternal and newborn health. At the Boston Consulting Group he led global health projects and contributed to projects on energy and the environment in both the public and private sectors.
Dr. Santos has a Ph.D. from Caltech, an A.B. from Vassar College, and was a postdoctoral fellow at the University of Cambridge and the Space Telescope Science Institute.