Reviewed by Danielle Ellis, B.Sc.Nov 6 2023
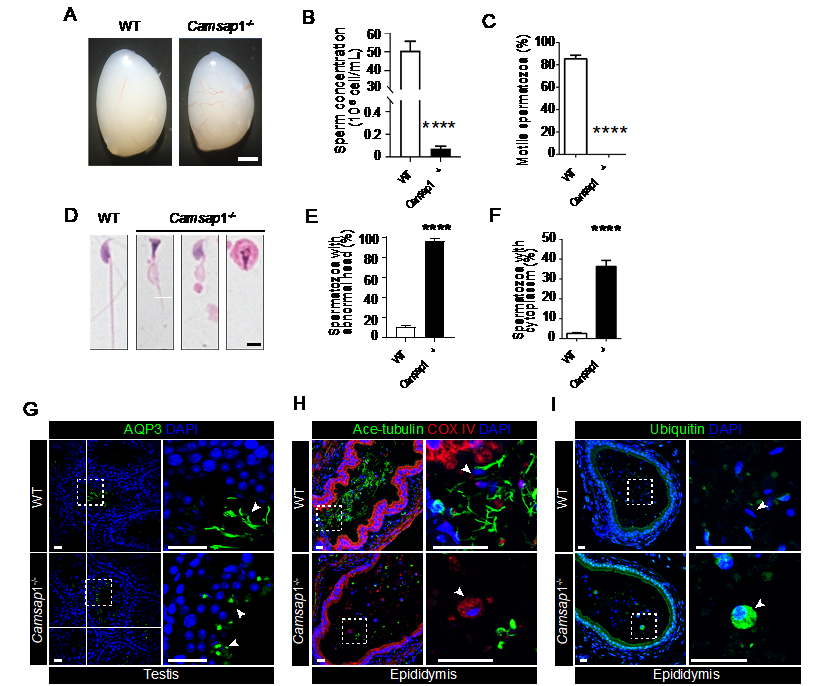
A team of researchers, led by Wenxiang Meng from the Institute of Genetics and Developmental Biology at the Chinese Academy of Sciences, has demonstrated that the deletion of the calmodulin-regulated spectrin-associated protein 1 (CAMSAP1) gene in mice, which is associated with the minus ends of non-centrosomal microtubules, results in a condition known as oligoasthenoteratozoospermia.
 Abnormal Sperm Development in CAMSAP1-/- Mice. Image Credit: IGDB
Abnormal Sperm Development in CAMSAP1-/- Mice. Image Credit: IGDB
The study was published in PNAS on October 30, 2023.
This condition encompasses various abnormalities in sperm, including atypical sperm head and tail morphology, reduced sperm count, diminished sperm motility, and male infertility.
During the process of sperm development, a transient structure called the manchette plays a crucial role.
The manchette primarily consists of non-centrosomal microtubules, and its proper regulation is essential for sperm formation. Disruption of these microtubules can lead to sperm abnormalities and, ultimately, male infertility.
Despite being discovered over half a century ago, little is known about the protein composition at the distal end of manchette microtubules and how it dynamically changes during sperm morphogenesis.
This study conducted histological and ultrastructural examinations of mouse testis and epididymis tissues, revealing that CAMSAP1 is localized to the distal end of manchette microtubules.
Image Credit: Anusorn Nakdee/Shutterstock.com
The absence of CAMSAP1 leads to abnormal elongation of manchette microtubules during spermatid shaping, a significant delay in the disassembly of CAMSAP1-deficient manchette microtubules, and an increase in acetylation modifications of microtubules.
Through the separation and enrichment of manchette components, proteomic analysis, and subsequent investigations, it was found that the loss of CAMSAP1 results in the abnormal localization of key proteins such as CEP170 and KIF2A at the distal end of the manchette. This disruption affects the anchoring of these proteins and delays the disassembly of manchette microtubules.
In essence, this study uncovers a novel mechanism for the dynamic regulation of manchette microtubules during sperm development. CAMSAP1 is shown to be instrumental in localizing CEP170 and KIF2A to the distal end of manchette microtubules, which in turn plays a critical role in anchoring and disassembling these microtubules.
This intricate process ensures the normal progression of sperm development and deepens our understanding of the causes of teratozoospermia.
Furthermore, this research provides a theoretical foundation for future clinical studies and therapeutic strategies for addressing teratozoospermia.
Source:
Journal reference:
Hu, W., et al. (2023). CAMSAP1 role in orchestrating structure and dynamics of manchette microtubule minus-ends impacts male fertility during spermiogenesis. Proceedings of the National Academy of Sciences of the United States of America. doi.org/10.1073/pnas.2313787120.