Automatic testing of a large number of biological and/or chemical compounds searching for a particular biological target is part of a drug discovery process called high-throughput screening (HTS).
These techniques are extensively used in the pharmaceutical sector, and utilize automation and robotics to test the biochemical or biological activity of several molecules — potential drugs — very rapidly.
Large-scale compound libraries can be rapidly screened in a cost-effective manner, thus accelerating target analysis. Moreover, high-throughput screening serves as a useful tool for assessing pharmacologically profiling agonists, pharmacological targets, and antagonists for enzymes and receptors (like GPCRs), for instance.
High-throughput screening in drug discovery
High-throughput screening is mainly performed to detect candidates that impact the target in a predicted manner, so-called “leads” or “hits” by screening compound libraries. This is achieved using plate readers as detectors, robotics, liquid handling devices, and dedicated software for data processing and instrumentation control.
High-throughput screening is not often used for drug identification: several properties that are important for the development of novel drugs cannot be assessed by compound library screening, like bioavailability and toxicity. Instead, the main function of high-throughput screening assays is to detect “leads” and provide suggestions to further improve them.
Assay results obtained from high-throughput screening serve as a preliminary point from which more stages in the drug discovery pipeline like drug design can be tracked. They also enable researchers to figure out the role or interaction of a specific biochemical process.
Therefore, high-throughput screening can be described as a process that allows rapid analysis of biological processes, through which compounds having a poor or no effect can be promptly excluded from the analysis pipeline.

PHERAstar FSX plate reader integrated in a Labcyte Access lab workstation at Cancer Research UK, Manchester Institute. Image Credit: BMG Labtech.
High-throughput screening services: Pharma, CROs, and Universities
The latest developments in liquid handling and plate reader detection, robotics, and also high-speed computers have led to the emergence of high-throughput screening. To run efficiently, high-throughput screening still requires an expensive and highly focused screening facility that cannot be afforded by all laboratories.
Rather than setting up their own facility, institutions with limited budgets routinely access high-throughput screening services through third-party providers like contract research organizations (CRO) or, particularly in core facilities and academia.
PHERAstar FSX supports the diverse and changing needs of CROs
Video Credit: BMG Labtech.
The high-throughput screening process: basic steps
From a technical standpoint, the major points in high-throughput screening are automation, miniaturization, and assay readout. High-throughput screening usually involves the following stages:
- Preparation of compound libraries and samples
- Formation of a robotic workstation
- Formation of a technique appropriate for automation
- Gaining and managing data
Preparation of samples and compound libraries
Usually, samples are biochemical or cellular in nature, based on the assay needed. In high-throughput screening, samples are prepared in an arrayed format — with microplate being the major platform.
Standard formats include 3456-, 1536-, or 384-well plates. The type of color and microplate format selected is probably governed by the nature of the detection assay and also by the nature of the sample. In screening facilities, compound library collections are preserved in what is known as “stock plates.” These are “copied” to an assay plate via a pipetting station when required, and are not directly utilized in experiments.
Establishment of a method suitable for automation
Automation is a major element of high-throughput screening, because the transition from a benchtop to an automated high-throughput screening assay enforces certain limitations which impact the practical design of the assay.
Ideally, a high-throughput screening assay is performed in a single well, requiring low concentrations of reagents (miniaturization) and minimal or no extra handling, except for injecting the compound or sample being examined. As a result, the choice of the assay and optimal detection mode has to be subordinated to automation problems.
Establishing an effective quality control technique and a rigorous assay are major problems when setting up an automated screen. If the distinction between positive and negative controls is clear, false negatives, particularly false positives, are less likely to be produced.
Configuration of a robotic workstation
The robotic platform has been developed to separately handle numerous plates at the same time, thus speeding up data acquisition considerably. For high-throughput screening, robotic platforms differ from basic automated liquid handling equipment to multidimensional workstations that perform various functions with the aid of one or more mechanical arms. Most often, a robotic system handles microplates from one station to another for a number of stages like addition, incubation, mixing, and detection of reagents.

CLARIOstar integrated in an automated biosynthetic production pipeline (Hamilton workstation), aimed at the production of fine and speciality chemicals from bacterial hosts at SYNBIOCHEM. Image Credit: BMG Labtech.
Acquisition and handling of data
An optical measurement is often used to perform data acquisition. This method enumerates the quantity of light “produced” by the sample. Different types of readouts like colorimetry, luminescent or fluorescent detection, or light scatter (turbidity) are provided.
FRET, fluorescence intensity and polarization, luminescence (for example, NanoBRET), time-resolved fluorescence (for example, LANCE, HTRF, etc.) and AlphaScreen are the standard detection modes.
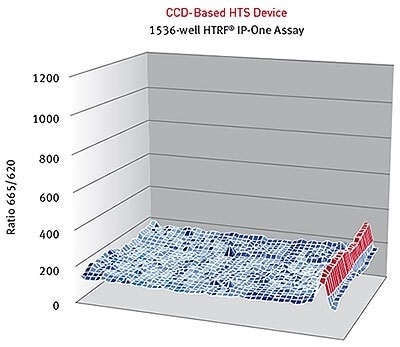
HTRF® ratios obtained for the IP-One assay with a CCD-based high-throughput screening device. Image Credit: BMG Labtech.
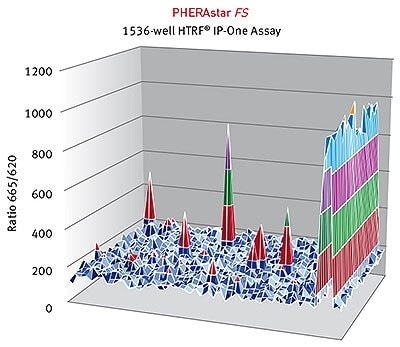
HTRF® ratios obtained for the IP-One assay with the PHERAstar FS. Image credits: BMG Labtech.
The light-based detection mode that is selected for readout relies on data quality, the biological question needed, and cost-effectiveness. Certain instrumentation, like multi-mode microplate readers, can apply varying detection procedures on the wells or successively perform different types of tests. A grid of numeric values is the output.

4 PHERAstar FSX plate readers integrated in a HighRes Biosolutions modular robotic workstation at AstraZeneca UK. Image Credit: BMG Labtech.
In just one day, a single HTS-dedicated plate reader can quantify an unlimited number of plates and create a huge amount of data points. Therefore, data management is crucial to automated HTS, considering the diversity of chemical libraries, a vast number of compounds tested, and the need to match and compare the outcomes from different types of screening operations.
Dedicated data analysis and management platforms (for example, Genedata) integrate the data acquired from various screening campaigns that have compound structures, apart from the completed assays, with each other. This helps in extracting complete data from different perspectives.
PHERAstar FSX: the dedicated high-throughput screening plate reader
It is important that compound library screenings are rapidly quantified in low volumes (a few µL) and produce robust results. Precise and sensitive instrumentation is required in addition to ease of use and the flexibility to perform different assays.
HTS on the PHERAstar
Video Credit: BMG Labtech.
The following reasons show why the PHERAstar FSX, through its past generations, is regarded as the gold standard reference reader for high-throughput screening:
- Concurrent dual emission detection for robust and fast detection of AlphaScreen assays, fluorescence polarization assays, TR-FRET, FRET, and BRET
- Option to quantify in 3456-, 1536- and 384-well plate formats
- Highest sensitivity for fluorescence polarization and intensity available on the market
- High-frequency TRF laser for efficient measurement of 1536-well plates on the go (1 flash) in only 36 seconds
- Dedicated AlphaLISA/AlphaScreen excitation laser
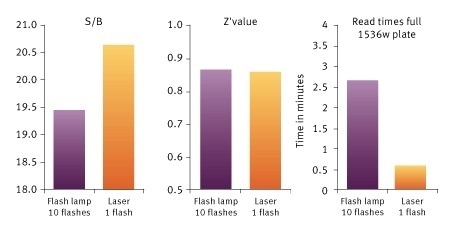
Signal to Blank (S/B), Z´value and read times for a Histone-BRD4 interaction assay measured on the PHERAstar FSX. The use of the TRF laser reduces read times, increases S/B, while keeping a very high Z´value. Image Credit: BMG Labtech.
High-Throughput Screening Applications
G-Protein Coupled Receptors, or GPCRs for short, are one of the most targeted receptor groups in drug screening. However, all bench-top assays cannot be changed into an automated HTS, particularly when live cells are being used.
The Application Note 287 of BMG Labtech describes for the first time how NanoBRET was effectively utilized to track ligand-GPCRs binding in living cells.
Miniaturization happens to be a major issue in HTS: samples should be very small to decrease the costs and still deliver reliable outcomes. The microplate reader’s sensitivity and its potential to reliably quantify samples in high-density plate formats are very significant.
Application Note 229 shows how DiscovereX PathHunter reporter cell lines were quantified using the PHERAstar FS microplate reader in both 3456- and 1536-well plates. The microplate reader detected a signal arising from just 1,000 cells in a 1,536-well plate and 250 cells in a 3,456-well plate.
In Application Note 260, the size of an AlphaLISA assay was reduced to 2 µL for each sample in a 1536-well plate for detection on the PHERAstar FS microplate reader.
Speed is as important as miniaturization in drug screening. Application Note 295 shows how the PHERAstar FSX plate reader fitted with the TRF excitation laser identifies HTRF assays in 1536-well plates “on the fly” in just 36 seconds, providing a Z` value of more than 0.8.
About BMG Labtech

BMG LABTECH has been committed to producing microplate readers for more than twenty years. By focusing on the needs of the scientific community, the company’s innovative microplate readers have earned the company the reputation of being a technology leader in the field.
BMG LABTECH has developed a wide range of dedicated and multi-mode microplate readers for life sciences applications and high-throughput screening.
All BMG LABTECH microplate readers are "Made in Germany" and are conceived, developed, assembled, and tested entirely at our headquarters in Germany.
Since our establishment in Offenburg, Germany in 1989, BMG LABTECH has expanded to offer a worldwide sales and support network with offices in the USA, UK, Australia, Japan and France. Our subsidiaries, regional offices and distributors are committed to bringing you innovative microplate reader technology with the quality and reliability you expect from a German company.
Our staff includes engineers and scientists from the fields of biology, biochemistry, analytical chemistry, and physics.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.