In the gene controlling mechanism, known as RNA interference, the expression of particular genes is downregulated by small interfering RNAs (siRNAs) or endogenic microRNAs.
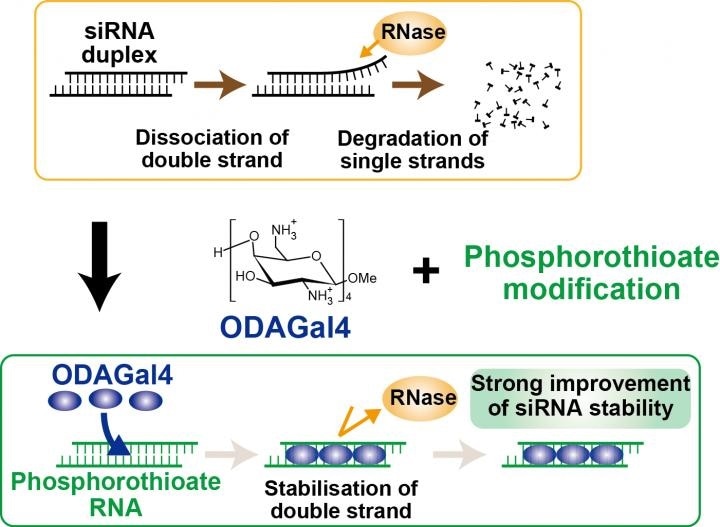
ODAGal4 strongly stabilizes duplex structures of siRNAs, particularly nucleotides with phosphorothioate linkages, and prevents degradation of the RNAs. Image Credit: Tokyo Metropolitan Institute of Medical Science.
siRNAs have extensive potential for gene-silencing therapy, but their instability poses a major challenge to create siRNA-based agents. Hence, to enhance their stability, a majority of the developed drugs based on siRNAs are chemically altered in their phosphodiester or nucleotides linkages. But chemical modification is not an ideal method for stabilizing siRNA because extensive alteration may disrupt the gene-silencing activity of siRNAs and also lead to cytotoxicity.
siRNAs contain oligonucleotide duplexes about 21-23 bases and create an A-form helical structure, wherein the major grooves have extremely negative potential, and hence, cationic molecules that can attach to these major grooves are anticipated to stabilize the RNA duplexes and guard them from cleavage in the body fluid.
On the basis of this concept, organic chemists from Tokyo University of Science have now produced an artificial cationic oligosaccharide, called oligodiaminogalactose 4mer (ODAGal4), that can preferentially attach to the major grooves of RNA duplexes to stabilize the siRNAs.
Atsushi Irie and his collaborator from Tokyo Metropolitan Institute of Medical Science in association with the research group at Tokyo University of Science have now devised a new method to stabilize the siRNAs using ODAGal4 together with phosphorothioate modification of RNAs.
In a study published online in the Scientific Reports journal on September 9th, 2020, the team has shown that ODAGal4 strongly improves the thermal and biological stability of siRNAs in vitro.
The team also showed that ODAGal4 has many special features for stabilizing the siRNAs. First, DAGal4 can enhance the stability of different siRNAs independent of the sequence of nucleotides, because ODAGal4 does not bind to the nucleobases of the nucleotides but instead attach to the phosphodiester linkages of RNA duplexes.
Most significantly, ODAGal4 does not affect the gene-silencing activity of any siRNAs. This feature of ODAGal4 is in sharp contrast to those of familiar chemical modifications, which may disrupt the gene-silencing activity of siRNAs. Thus, ODAGal4 has excellent potential for siRNA stabilization, as it is extensively relevant to numerous siRNA-based drugs.
Second, the impact of ODAGal4 on the stabilization of siRNAs is further improved by chemical alteration of the siRNAs; Specifically, ODAGal4 profoundly enhances the stability of RNAs with phosphorothioate linkages. This enhanced stability of siRNA is superior to that seen for other chemical modifications (for example, 2'-deoxy- 2'-fluoro nucleotides and 2'-O-methyl, locked nucleic acid) indicating that ODAGal4 integrated with phosphorothioate modification is extremely effective for siRNA stabilization.
Finally, another striking characteristic of ODAGal4 is its binding specificity to duplexes of RNAs; ODAGal4 attaches to A-form RNA helix but neither to B-form DNA helix nor to single-stranded DNA/RNA.
While numerous gene delivery systems containing cationic polymers have been designed to stabilize nucleotides, the molecular structures of the polymers are not developed to particularly attach to nucleotides.
The binding between the polymers and the nucleotides depends on the ionic interaction that exists between them, and hence, the polycation complexes are likely to cause cytotoxicity because of nonspecific binding of the polymers to other kinds of biomolecules. In sharp contrast, ODAGal4 does not cause cytotoxicity due to its limited binding with high affinity to RNA duplexes.
Our goal will be application of ODAGal4 for siRNA-based agents. Although we should study in vivo experiments to confirm and expand our findings, we emphasise that ODAGal4 has a great advantage as improving siRNA stability and has a potential for reducing total dose and frequency of administration of siRNA-based drugs in future application.”
Atsushi Irie, Tokyo Metropolitan Institute of Medical Science
Source:
Journal reference:
Irie, A., et al. (2020) An artificial cationic oligosaccharide combined with phosphorothioate linkages strongly improves siRNA stability. Scientific Reports. doi.org/10.1038/s41598-020-71896-w.