Zika outbreaks that occurred back in 2015 and 2016 are still having lasting effects on children whose mothers were infected with the Zika virus during their pregnancy.
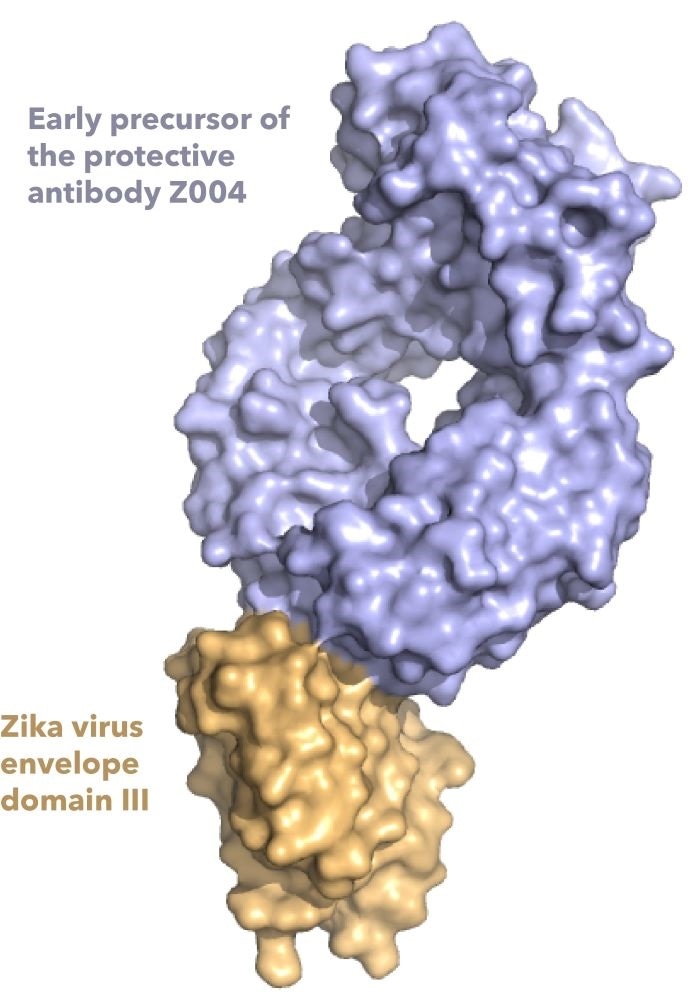
A structure of an antibody, called Z004 (shown in purple), bound to the envelope domain III protein of Zika virus (shown in gold). Image Credit: Shannon Esswein.
While the number of infections of the Zika virus has declined, which researchers believe may be due to herd immunity in certain regions, there is still a risk of future outbreaks. Hence, to stop such outbreaks, researchers want to learn how the Zika virus is recognized by the immune system, in hopes of designing vaccines against this pathogen.
Shannon Esswein, a graduate student, and Pamela Bjorkman, a professor from the California Institute of Technology, have gained a new understanding of how the antibodies in the body bind to the Zika virus. Esswein will present the study at the 65th Biophysical Society Annual Meeting on Thursday, February 25th, 2021. The study was published in the PNAS journal.
Zika virus is a type of flavivirus, and other family members of flavivirus comprise dengue, yellow fever, and West Nile viruses.
To protect from these and other similar pathogens, “We have the ability to make a huge diversity of antibodies, and if we get infected or vaccinated, those antibodies recognize the pathogen,” stated Esswein.
But at times, when the body sets off an immune reaction against a flavivirus, it is believed that this response could make the individuals sicker if they get infected the next time.
Dubbed antibody-dependent enhancement (ADE), occurs when the antibodies bind to the exterior part of the virus without inhibiting its potential to infect cells and can unintentionally help the virus to infect more numbers of cells by enabling this pathogen to penetrate the cells to which the antibodies are bound.
To prevent ADE when designing a vaccine, researchers should have a thorough understanding of how antibodies bind to a particular virus. This is particularly significant for flaviviruses, because antibodies that defend against a single flavivirus are also likely to stick to, but not protect against, other flaviviruses, thus raising the risk of ADE.
There are some concerns that antibodies produced in response to a Zika virus vaccine could activate ADE if some person were to be later infected with various flaviviruses, including dengue.
Esswein and Bjorkman looked at multiple blood antibodies to analyze the antibody response to various flaviviruses, including Zika, These antibodies were collected from the blood of patients in Brazil and Mexico.
To identify antibodies that detect flaviviruses, the team utilized a part of the outside of the virus, known as the envelope domain III protein. Prior studies have demonstrated that the envelope domain III is a significant target of protective antibodies that combat flavivirus infections.
The research team examined how these antibodies transformed over time as they develop and become better at binding to the Zika virus, and also how antibodies are able to cross-react with other flaviviruses, such as the four forms of dengue viruses. The researchers also observed that the Zika antibodies tightly bind to and protect against dengue type 1 but weakly bind to dengue type 2 and 4 and West Nile viruses.
The weak cross-reactivity of these antibodies doesn’t seem to defend against those flaviviruses, but also doesn’t induce ADE, suggesting envelope domain III may be useful to make a vaccine that is safe.”
Shannon Esswein, Graduate Student, California Institute of Technology
The team also established structures, demonstrating how Zika and West Nile envelope domain III are identified by two antibodies.
Together, the researchers’ experiments demonstrate how the body triggers “a potent immune response to Zika virus,” concluded Esswein. the researchers’ insights into the antibodies that played a role in this immune reaction will help inform new design strategies for vaccines.
Source:
Journal reference:
Esswein, S. R., et al. (2021) Structural basis for Zika envelope domain III recognition by a germline version of a recurrent neutralizing antibody. PNAS. doi.org/10.1073/pnas.1919269117.