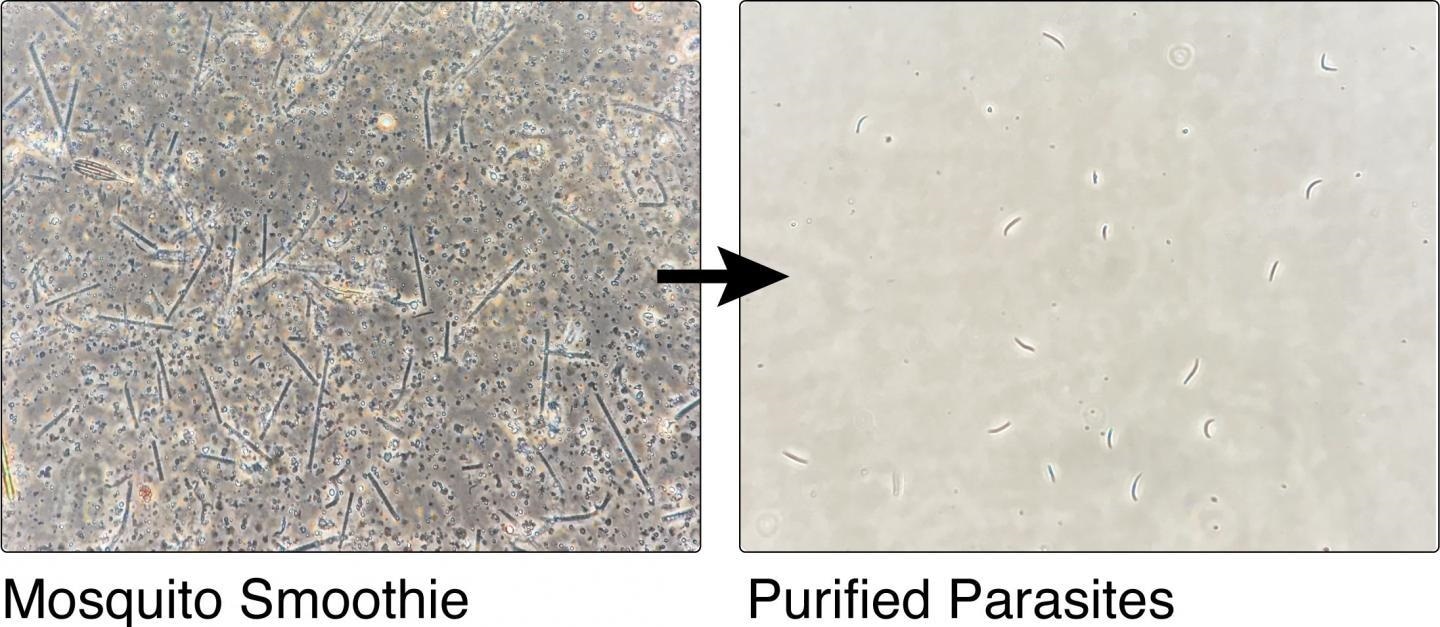
The mosquito 'smoothie' and the purified parasites after the team's process. Image Credit: Joshua Blight/Imperial College London.
Malaria parasites are becoming increasingly resistant to antimalarial medicines, while mosquitos that spread the disease are becoming more and more immune to insecticides. This has created an urgent need to identify innovative ways to combat malaria, the world’s third-deadliest disease in children under the age of five, with one child dying from malaria every two minutes.
Present-day malaria vaccines that employ the whole parasites offer only modest protection against the disease. The parasites in these vaccines are “attenuated,” similar to certain flu vaccines and the MMR vaccine, such that they infect individuals and trigger a powerful immune response that protects them against malaria, but does not cause the disease themselves.
But these vaccines need multiple doses, with each injection possibly requiring tens of thousands of parasites at an early stage of growth, called sporozoites. Sporozoites are typically present in the mosquito’s salivary glands, and in a natural infection, are transmitted to humans when the mosquito bites. They subsequently spread to the human liver, where they prepare to infect the body.
To extract sporozoites for use in a live vaccine, a skilled technician must manually dissect the mosquito salivary glands (tiny structures located behind the mosquito head)—a process that is both costly and involves a great deal of time.
No dissection required
The new approach, recently published in the Life Science Alliance journal, significantly accelerates this process by effectively producing a “mosquito smoothie’ and subsequently filtering the resultant liquid by density, size, and electrical charge. This process yields a pure sporozoite product that is best suited for vaccination. Most significantly, there is no need for dissection.
Creating whole-parasites vaccines in large enough volumes and in a timely and cost-effective way has been a major roadblock for advancing malaria vaccinology, unless you can employ an army of skilled mosquito dissectors. Our new method presents a way to radically cheapen, speed up and improve vaccine production.”
Jake Baum, Lead Researcher and Professor, Department of Life Sciences, Imperial College London
But it is not just about cost and speed. Conventional dissection procedures generally struggle to eliminate all pollutants, like proteins from the salivary glands, which are frequently removed with sporozoites. The additional debris may probably influence sporozoite’s infectivity once these pathogens are inside the body, and may even influence the response of the immune system, thus affecting the efficacy of any whole parasite vaccine.
The new technique also addresses this concern, leading to pure, uncontaminated samples of sporozoites. The researchers also observed that in addition to being purer, the resultant sporozoites were unexpectedly more infectious, indicating that vaccines produced through the new technique may need a relatively lower dose of sporozoites.
With this new approach we not only improve the scalability of vaccine production, but our isolated sporozoites may actually prove to be more potent as a vaccine, giving us additional bang per mosquito buck.”
Dr Joshua Blight, Study First Author, Department of Life Sciences, Imperial College London
Towards new vaccines
The researchers devised and tested the new technique with both rodent and human malaria parasites. They later tested the rodent version as a vaccine in mice and noted that when vaccinated mice are subjected to an infected mosquito bite, they displayed a 60% to 70% protection when immunizations were administered into the muscle. But when the same set of sporozoites were administered directly into the bloodstream (intravenously), protection was 100%, a condition known as “sterile” protection.
The team is currently refining the process to prepare for large-scale production of sporozoites under good manufacturing practice (GMP) conditions to create a vaccine set for human challenge trials. Under this plan, participants would receive vaccine-grade sporozoites generated using the new method before they are deliberately bitten by an infected mosquito.
In addition to vaccinations, the researchers believe that this new technique will help expedite studies of sporozoite biology in general, which may ultimately lead to new insights into the liver stage of malaria as well as novel drug and vaccine regimens.
Source:
Journal reference:
Blight, J., et al. (2021) Dissection-independent production of Plasmodium sporozoites from whole mosquitoes. Life Science Alliance. doi.org/10.26508/lsa.202101094.