A new study has discovered a two-arm molecule that can effectively deplete cancer-protecting cells within tumors, which thus enables the immune system to combat tumors without turning overactive.
The binding of CTLA-4 antibodies throughout the body limits their anti-tumor effect. In contrast, a newly developed two-armed molecule that targets both CLTA-4 and CD47 on regulatory T cells efficiently hones in on tumors and enhances antitumor immunity. Image Credit: University of Texas Southwestern Medical School.
Published online in Science Translational Medicine, the results of the study could pave the way for new types of cancer immunotherapies.
By effectively removing these immune-suppressive cells inside tumors instead of in the whole body, the immune system can attack cancers without causing harmful autoimmune conditions.”
Yang-Xin Fu, MD, PhD, Study Leader and Professor of Pathology, Immunology, and Radiation Oncology, UT Southwestern
Dr. Fu co-led this study with Anli Zhang, Ph.D., and Zhenhua Ren, Ph.D., two postdoctoral researchers from his lab.
For several years, scientists have been aware that the immune system not just has a crucial role in fighting off cancers through the direct action of killer T cells and other components. It also resists these efforts through cells called regulatory T cells, or Tregs.
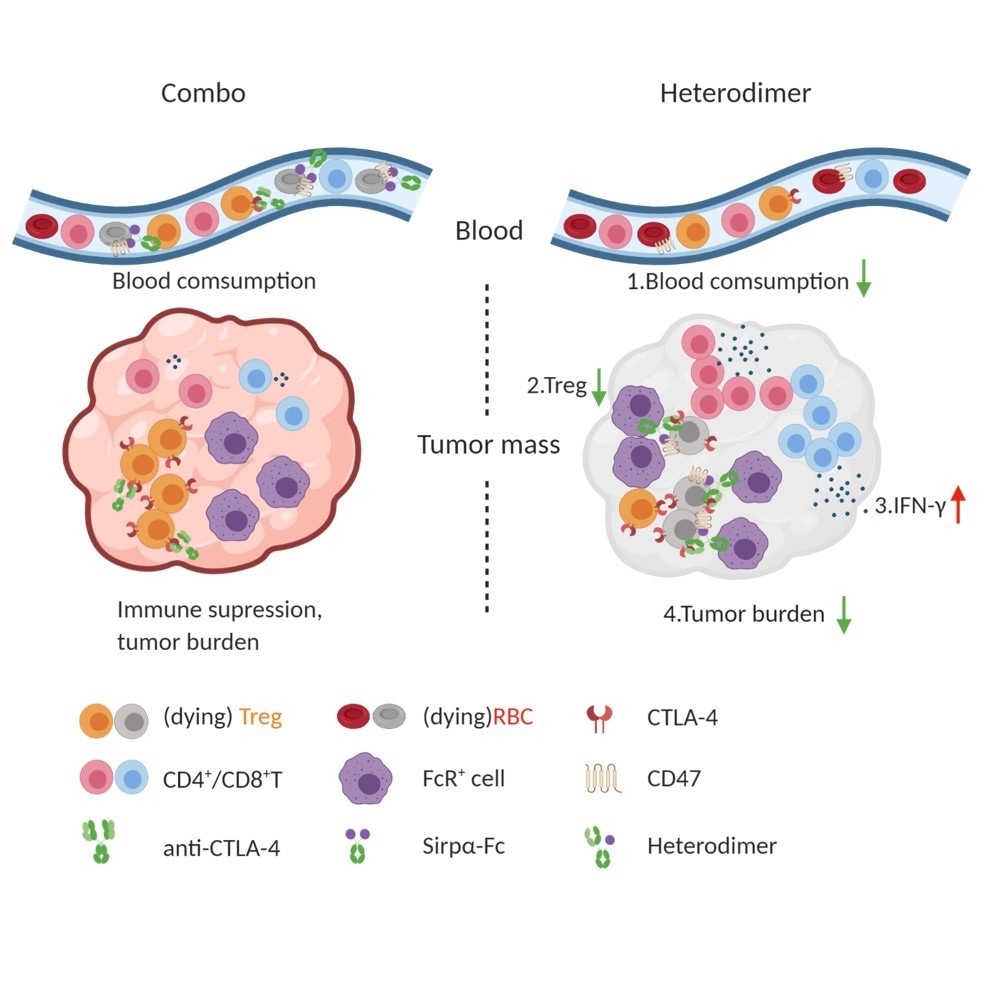
The Tregs enable the immune response to be regulated by inhibiting different immune cells from turning overactive and causing autoimmune diseases. But they also accumulate in tumors, protecting them from immune attack.
According to Dr. Fu, Tregs preserve a balance of two proteins on their surfaces—CTLA-4 and CD47—that respectively transmit “eat me” and “don’t eat me” signals to phagocytes that control Tregs. Several immunotherapies have aimed to boost the “eat me” signal or reduce the “don’t eat me signal” to decrease Tregs in tumors.
But each approach has its own disadvantage: An increase in the “eat me” signal causes systemic effects that drive autoimmunity, and a decrease in the “don’t eat me” signal has only shown potential for the treatment of blood cancers, like leukemias.
Drs Fu, Zhang, Ren, and their collaborators looked for a new way to deplete Tregs and thus developed a two-arm molecule that increases the “eat me” signal while also inhibiting the “don’t eat me” signal to stimulate phagocytes to devour those immune suppressive cells.
Upon injecting into mouse models of colon cancer, the molecule was found to preferentially deplete Tregs in tumors without any impact on those in the rest of the body, thereby sparing the animals from treatment-driven autoimmune disease.
But when the animals were dosed with equivalent, individual amounts of the “eat me” booster and “don’t eat me” blocker, there were systemic autoimmune side effects, indicating that blending them within one molecule is crucial to reaching Tregs in tumors.
With the decrease in the number of Tregs as part of treatment, the tumors in the animals shrank considerably. This approach also worked in mice affected by human lung cancer tumors, implying that it could be feasible in human patients.
In the past, there’s been no way to get rid of these suppressive Tregs without severe toxicity. Our study suggests a way to this outcome.”
Yang-Xin Fu, MD, PhD, Study Leader and Professor of Pathology, Immunology, and Radiation Oncology, UT Southwestern
Source:
Journal reference:
Zhang, A., et al. (2021) Dual targeting of CTLA-4 and CD47 on Treg cells promotes immunity against solid tumors. Science Translational Medicine. doi.org/10.1126/scitranslmed.abg8693.