Scientists from the group of Hans Clevers in association with the group of Bart Haagmans (Erasmus MC) created an organoid biobank to investigate the genes that are vital for the spreading of a SARS-CoV-2 infection.
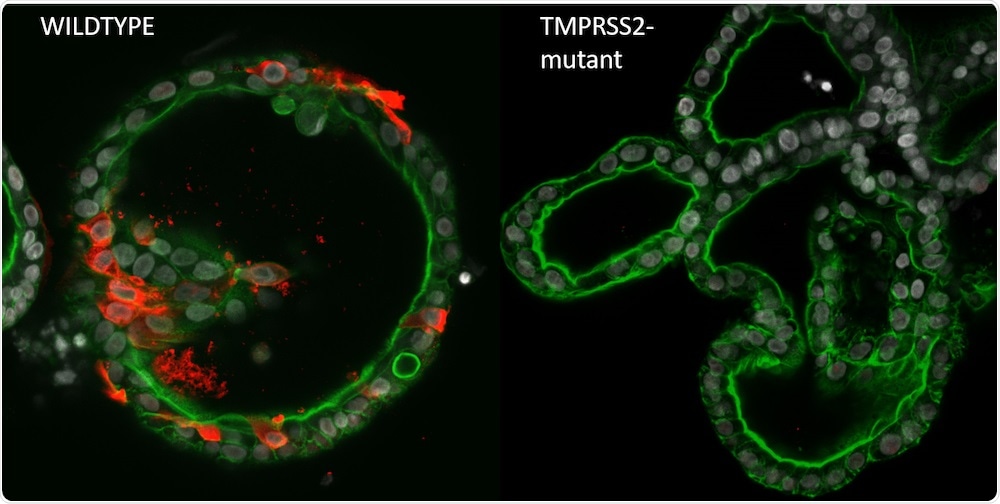
SARS-CoV-2 (indicated by nucleocapsid, red) efficiently infects wildtype human intestinal organoids (left), while TMPRSS2-deficient organoids are completely devoid of virus (right). Nuclei are grey, phalloidin (green) visualizes actin filaments. Image Credit: Joep Beumer, copyright Hubrecht Institute.
The research outlines the advantages of organoids for fundamental research into coronaviruses, along with emphasizing potential drug targets. The study was published on September 17th, 2021, in the Nature Communications journal.
Organoids are small 3D structures grown from stem cells that mirror organ function. Scientists have earlier successfully created organoid models for numerous organs including the lung, uterus, gut, and even the snake venom gland. The organoids are proved to be beneficial for foretelling therapy responses in patients, diagnostic purposes, and revealing secrets about the development of tissues and rare cell types.
Host factor
Scientists can introduce levels of complexity to the organoid cultures for specific purposes. They can, for instance, inject pathogens into organoids to model their effect on the cells or introduce immune cells to tumor organoids to investigate the efficacy of therapy. The former approach is applied currently to model coronavirus infections in human cells.
The major question with coronavirus—and viruses as a whole—is about the factors it utilizes to penetrate human cells and replicate. These alleged host factors could be fascinating drug targets to affect viral replication and spreading.
Biobank of intestinal organoids
To unearth more information about the host factors vital for the replication and spreading of coronaviruses, the teams from Hans Clevers and Bart Haagmans created a biobank of mutant intestinal organoids.
This indicates that the organoids, that mirror the biology of the intestine, included numerous mutations in the host factors that were earlier found to be relevant for coronaviruses. These mutations induce changes in the activity of the host factors.
TMPRSS2 as a therapeutic target
Subsequently, the scientists injected SARS-CoV-2—the virus responsible for COVID-19—into the mutant organoids to investigate the impact of the mutations on the replication and spreading of the virus. In addition to other factors, they pinpointed the gene TMPRSS2 to be related to this mechanism: organoids with non-functioning TMPRSS2 displayed decreased replication and spreading of the virus.
Hence, this gene might be an attractive therapeutic target for the coronavirus. Recently, specific inhibitors for TMPRSS2 have been created.
Relevance of human models
Earlier research employed cell lines of animals (significantly of the African green monkey) to pinpoint therapeutic targets for the coronavirus. The cell lines were effortless to work with; however, they do not completely recapitulate the biology of human cells that are targets of SARS-CoV-2.
This is depicted by the anti-malaria drug Chloroquine, which was identified to be efficient against SARS-CoV-2 infection in these cell lines but proved to be inefficient in clinical trials with patients. This demonstrates that the cell lines cannot adequately foretell the efficacy of therapeutics in humans.
While the experiments with Chloroquine were repeated by employing the mutant organoids rather than earlier employed cell lines, the scientists found no therapeutic effect. Alternatively stated, the results observed in organoids were identical to the results from clinical trials, implying that—when compared to animal cell lines—the organoids would better suit to foretell the efficacy of therapeutics in humans.
Future viruses
The research teams from Hans Clevers and Bart Haagmans emphasize the importance of organoids for research into coronaviruses. Moreover, they pinpoint TMPSS2 as a possible therapeutic target for SARS-CoV-2. The recently created biobank can also help screen future emerging viruses to quickly identify therapeutic targets.
Source:
Journal reference:
Beumer, J., et al. (2021) A CRISPR/Cas9 genetically engineered organoid biobank reveals essential host factors for coronaviruses. Nature Communications. doi.org/10.1038/s41467-021-25729-7.