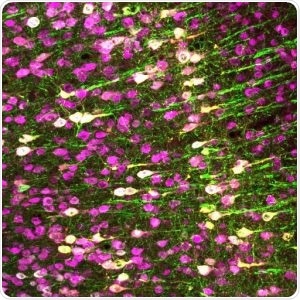
Neurons (magenta) in the mouse brain. The Chalasani lab made specific neurons express TRPA1 (white), so they can be activated by ultrasound. Image Credit: Salk Institute for Biological Studies.
Chalasani established the idea of employing ultrasonic waves to activate particular groups of genetically marked cells, about a decade ago. To describe it, he devised the term “sonogenetics.”
His team showed in 2015 that in Caenorhabditis elegans (roundworm)—a protein called TRP-4—makes cells sensitive to low-frequency ultrasound. When the scientists added TRP-4 to C. elegans neurons, where the protein is usually absent, they could stimulate these cells with a spurt of ultrasound—similar to the sound waves utilized in medical sonograms.
However, when the scientists tried adding TRP-4 to mammalian cells, the protein was unable to make the cells respond to ultrasound. Although some mammalian proteins were stated to be ultrasound-sensitive, none appeared to be ideal for clinical use. Hence, Chalasani and his co-researchers set out to look for a new mammalian protein that made cells extremely sensitive to ultrasound at 7 MHz that is believed to be an optimal and safe frequency.
Our approach was different than previous screens because we set out to look for ultrasound-sensitive channels in a comprehensive way.”
Yusuf Tufail, Study Co-First Author and Former Project Scientist, Salk Institute for Biological Studies
The scientists added hundreds of diverse proteins, all at one time, to a common human research cell line (HEK) that is usually insensitive to ultrasound. Further, they placed each cell culture beneath a setup that allowed them to monitor variations to the cells upon ultrasound activation.
Finally, the scientists found a protein that made the HEK cells responsive to the 7 MHz ultrasound frequency after screening proteins for over a year and working their way across around 300 candidates. A channel protein, called TRPA1, was found to allow cells to respond to the existence of noxious compounds and to stimulate a set of cells in the human body, alongside heart and brain cells.
But Chalasani’s team found that the protein channel was also responsive to ultrasound in HEK cells.
Marc Duque, a co-first author of the paper and Salk exchange student, says “We were really surprised. TRPA1 has been well-studied in the literature but hasn’t been described as a classical mechanosensitive protein that you’d expect to respond to ultrasound.”
The team employed a gene therapy method to add the genes for human TRPA1 to a particular group of neurons in the brains of living mice to verify whether the channel could stimulate other cell types as a response to ultrasound. Only the neurons with the TRPA1 genes were stimulated when the team administered ultrasound to the mice.
Currently, the medical treatments of conditions such as Parkinson’s disease and epilepsy use deep brain stimulation, involving surgically implantation of electrodes in the brain, to stimulate particular subsets of neurons. Chalasani says that this approach can be replaced by sonogenetics one day—where a step forward would be building a gene therapy delivery method that can surpass the blood-brain barrier, which has been already studied.
He adds that sonogenetics can be employed to stimulate cells in the heart, like a type of pacemaker that needs no implantation.
Sreekanth Chalasani states, “Gene delivery techniques already exist for getting a new gene—such as TRPA1—into the human heart. If we can then use an external ultrasound device to activate those cells, that could really revolutionize pacemakers.”
Currently, his team is executing more fundamental work on how exactly TRPA1 senses ultrasound.
In order to make this finding more useful for future research and clinical applications, we hope to determine exactly what parts of TRPA1 contribute to its ultrasound sensitivity and tweak them to enhance this sensitivity.”
Corinne Lee-Kubli, Study Co-First Author and Former Postdoctoral Fellow, Salk Institute for Biological Studies
For ultrasound-sensitive proteins, the team also intends to perform another screen, which would look for proteins that can hinder, or shut off, a cell’s activity as a response to ultrasound.
Progress for Sonogenetics a non-invasive way to treat brain disorders
Video Credit: Salk Institute for Biological Studies.
Source:
Journal reference:
Duque, M., et al (2022) Sonogenetic control of mammalian cells using exogenous Transient Receptor Potential A1 channels. Nature Communications. doi.org/10.1038/s41467-022-28205-y