Reviewed by Danielle Ellis, B.Sc.Jun 6 2022
As the efficiency of conventional antimalarial medications deteriorates, a novel approach to combating malaria that sees the disease turn against itself could provide a viable treatment for the hundreds of millions of people affected globally every year.
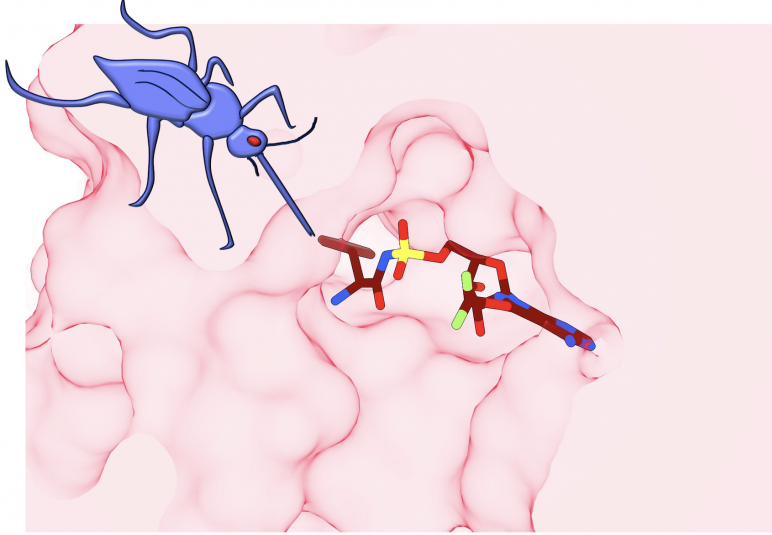 Diagrammatic representation of the target of the antimalarial compound ML901 (colored structure), showing highly specific and potent inhibition of the malaria parasite. ML901 finds a particular chink in an enzyme called tyrosine tRNA synthetase (depicted in pink), part of the machinery that the malaria parasite uses to generate the proteins needed to reproduce itself. The parasite rapidly grinds to a halt and can’t cause disease or be transmitted to other people via mosquitoes (purple). Image Credit: Leann Tilley and Riley Metcalfe
Diagrammatic representation of the target of the antimalarial compound ML901 (colored structure), showing highly specific and potent inhibition of the malaria parasite. ML901 finds a particular chink in an enzyme called tyrosine tRNA synthetase (depicted in pink), part of the machinery that the malaria parasite uses to generate the proteins needed to reproduce itself. The parasite rapidly grinds to a halt and can’t cause disease or be transmitted to other people via mosquitoes (purple). Image Credit: Leann Tilley and Riley Metcalfe
ML901, an anti-malarial chemical discovered by researchers at the University of Melbourne and published in Science, suppresses the malaria parasite without harming mammalian—human or other mammals’—cells.
Professor Leann Tilley of the University’s Bio21 Institute, who was a co-lead author on the study, said the ML901 molecule effectively made the parasite the agent of its destruction, demonstrating its strength and specificity.
ML901 works by an unusual reaction-hijacking mechanism.”
Leann Tilley, Study Co-Lead Author and Professor, Bio21 Institute, University of Melbourne
Tilley also stated, “Imagine a stealth weapon that can be used to launch a self-destruct attack on your vehicle—slamming on the brakes and cutting the engine. ML901 finds a particular chink in the machinery that the malaria parasite uses to generate the proteins needed to reproduce itself and stops it from doing so.”
“While there is much work to be done to fine-tune what we’ve discovered, these results are encouraging in the search for new antimalarials,” Tilley added.
Tests were undertaken utilizing molecules given by Takeda in partnership with Takeda Pharmaceuticals, Medicines for Malaria Medicine—the peak international group for antimalarial drug development—and research facilities across five continents, during which the ML901 chemical was discovered.
Once inside the parasite, ML901 hooked itself to an amino acid and destroyed the parasite’s protein synthesis machinery from within, crushing it to a standstill. Human cells are not vulnerable to ML901 because of their molecular structure.
The scientists discovered that ML901 killed malaria parasites that were resistant to presently used medications in studies using both human blood cultures and an animal model of malaria. It also demonstrated rapid and persistent activity, resulting in excellent parasite killing.
Professor Tilley stated that the molecule was active against all phases of the lifecycle, implying that it may be used to both prevent and treat malaria infections.
It also shows potential for preventing infected people from transmitting the disease to others, which is critical to stop the spread of malaria.”
Leann Tilley, Study Co-Lead Author and Professor, Bio21 Institute, University of Melbourne
At least 200 million new malaria infections are identified each year around the world, resulting in over 600,000 fatalities in Africa and Southeast Asia. Antimalarial resistance has risen steadily over the last 50 years, signaling an imminent catastrophe in which breakthrough treatments are sorely needed.
Professor Tilley stated that the team was prepared to pursue the development of novel antimalarial medication candidates based on these results.
We believe this is just the beginning. We now have the possibility of finding drugs, similar to ML901, that target a range of deadly infectious diseases, including multi-drug resistant bacterial infections. The work opens up several new drug discovery avenues.”
Leann Tilley, Study Co-Lead Author and Professor, Bio21 Institute, University of Melbourne
Source:
Journal reference:
Xie, S. C., et al. (2022) Reaction hijacking of tyrosine tRNA synthetase as a new whole-of-life-cycle antimalarial strategy. Science. doi.org/10.1126/science.abn0611.