Reviewed by Danielle Ellis, B.Sc.Jun 19 2023
Malaria continues to be a serious public health issue, especially in sub-Saharan Africa, with almost 250 million cases per year and 621,000 fatalities. The parasitic disease malaria is spread by mosquitoes and is caused by a microbe of the genus called Plasmodium.
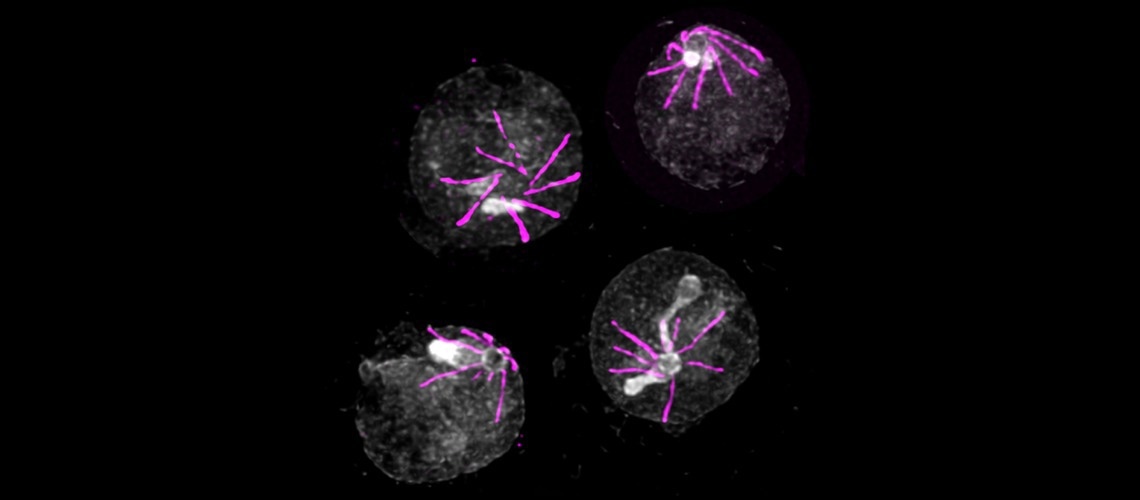 Form of Plasmodium responsible for the destruction of red blood cells as seen by expansion microscopy. Image Credit: Vincent Louvel et Eloïse Bertiaux
Form of Plasmodium responsible for the destruction of red blood cells as seen by expansion microscopy. Image Credit: Vincent Louvel et Eloïse Bertiaux
Plasmodium must adjust to the unique characteristics of the several organs and cells it parasitizes during its trip from mosquito to person. Microbes lack sensory organs; instead, they use protein-based sensors to find molecules unique to the surroundings they inhabit.
Plasmodium is an exception to the rule that most living creatures have the same types of sensors. University of Geneva (UNIGE) biologists have discovered a new form of sensor that allows Plasmodium to know exactly where it is and what to do.
This study, published in the journal Science Advances, suggests that scrambling the signals seen by this sensor could be used to disorient the parasite and hence impede its replication and proliferation.
When a Plasmodium-infected mosquito bites a human, the parasite enters the bloodstream and travels to the liver, where it thrives for about ten days without generating any symptoms. Plasmodium then re-enters the bloodstream, where it parasitizes red blood cells. The parasites replicate in a synchronized 48-hour cycle once within the red blood cells.
Image Credit: Kateryna Kon/Shutterstock.com
The newly created parasites leave their host red blood cells after each cycle of multiplication, killing them and infecting fresh ones. The waves of fever linked to malaria are caused by this loss of red blood cells. The blockage of blood vessels by infected red blood cells has been associated with severe forms of malaria.
When a mosquito bites a person whose blood contains Plasmodium, the parasite modifies its course of development to colonize its new host’s intestine. Plasmodium returns to the salivary glands of the mosquito after a second round of replication, prepared to infect a fresh victim.
Unknown Communication Channels
How can Plasmodium sense changes in its surroundings to alter its development plan, from the warmth of the red blood cell to the depths of the intestines of the mosquito via the liver?
Understanding this very specific biological mechanism is an important step towards countering the parasite. At each stage of its life cycle, the parasite must logically pick up signals that enable it to react correctly, but which ones and how?"
Mathieu Brochet, Associate Professor, Department of Microbiology and Molecular Medicine, Faculty of Medicine, University of Geneva
The parasite is able to recognize certain small molecules that are present in the mosquito but absent from the blood.
Ronja Kühnel and Emma Ganga, PhD students in Mathieu Brochet’s laboratory and first authors of this study, added, “Starting from this single known element, we have identified a sensor that enables the parasite to detect the presence of these molecules when it is ingested by a mosquito. This sensor is made up of five proteins. In its absence, the parasite does not realize that it has left the bloodstream for the mosquito, and is therefore unable to continue its development.”
Surprisingly, this sensor is also active during other phases of the life cycle of the parasite, particularly when it is time for the parasite to depart the red blood cell.
Kühnel and Ganga further stated, “We then observe exactly the same mechanism: without this sensor, Plasmodium is trapped in the red blood cells, unable to continue its infection cycle.”
Scientists have not identified the human molecules recognized by the parasite; finding them could provide a better knowledge of how Plasmodium causes fever waves.
Other Parasites Also Involved
The protein complex identified here is lacking in humans but is present in Toxoplasma, the causative agent of toxoplasmosis, as well as the entire family of apicomplexan parasites to which Plasmodium belongs.
The discovery of this sensor has allowed researchers to speculate on ways to confuse the signals the parasite perceives at various phases of its development to disorient it and prevent its transmission and multiplication.
Source:
Journal reference:
Kühnel, R. M., et al. (2023). A Plasmodium membrane receptor platform integrates cues for egress and invasion in blood forms and activation of transmission stages. Science Advances. doi.org/10.1126/sciadv.adf2161