Reviewed by Danielle Ellis, B.Sc.Oct 26 2023
Scientists from Stockholm University have revealed the concealed complexities behind the transition of sperm from inert observers to active swimmers.
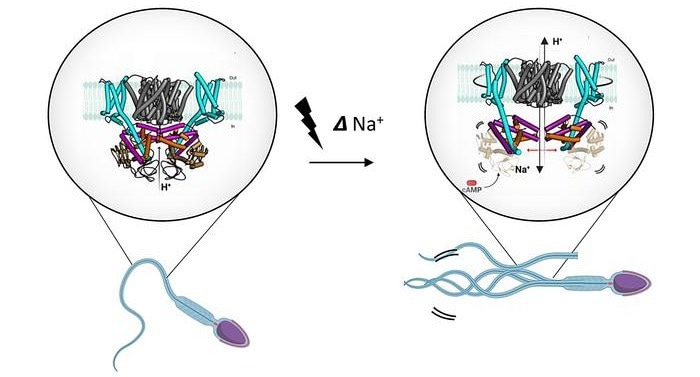 Chemo-attractant binding to sperm causes a change in voltage, which activates the transporter SLC9C1. The structure of SLC9C1 is here described for the first time. Once activated the SLC9C1 protein exchanges Na+ for H+ ions and this makes the sperm flagellum more alkaline and, together with cAMP, leads to the opening of Ca2+-channels that result in a directed movement of the sperm. This sequence of events, present in species as distant as corals and human, are essential for fertilization. Image Credit: Ved Mehta/Stockholm University.
Chemo-attractant binding to sperm causes a change in voltage, which activates the transporter SLC9C1. The structure of SLC9C1 is here described for the first time. Once activated the SLC9C1 protein exchanges Na+ for H+ ions and this makes the sperm flagellum more alkaline and, together with cAMP, leads to the opening of Ca2+-channels that result in a directed movement of the sperm. This sequence of events, present in species as distant as corals and human, are essential for fertilization. Image Credit: Ved Mehta/Stockholm University.
This metamorphosis is a crucial stage in the path to fertilization, relying on the activation of a distinctive ion transporter.
Picture sperm as minuscule explorers embarking on a quest to find the ultimate treasure—the egg. Without a map, they rely on something even more remarkable: chemo-attractants. These are chemical cues emitted by the egg, serving as a mesmerizing beacon that guides and energizes the sperm.
When these cues bind to receptors on the sperm's exterior, it initiates a cascade of actions, setting them on the path toward the egg. In this intricate narrative, a pivotal character is a protein named “SLC9C1.”
Image Credit: ninoon/Shutterstock.com
This protein, “SLC9C1,” is exclusively located in sperm cells and typically remains dormant. Nonetheless, when the chemo-attractants engage with the sperm's surface, a remarkable transformation occurs.
SLC9C1 operates like a highly sophisticated exchange system. It swaps protons from inside the cell for sodium ions from the outside, temporarily creating a less acidic environment within the sperm. This change in the internal environment triggers increased sperm motility.”
David Drew, Professor, Biochemistry, Stockholm University
The activation of SLC9C1 links on a voltage shift triggered when chemo-attractants bind to the sperm. To achieve this, SLC9C1 employs a distinctive feature known as a voltage-sensing domain (VSD). Normally, VSD domains are linked to voltage-gated ion channels. However, in the case of SLC9C1, it stands out as truly exceptional in the realm of transporters.
Under the guidance of David Drew, a team of researchers has unveiled the inner workings of SLC9C1, offering the initial instance of voltage-sensing domain activation in a transporter, and its association through an unusually elongated voltage-sensing (S4) helix.
The VSD domain responds to the change in voltage by pushing its rodlike S4 helix inwards. This clears the way for ion exchange by SLC9C1, ultimately initiating sperm motility.”
David Drew, Professor, Biochemistry, Stockholm University
Drew added, “Transporters work very differently than channels and, as such, the VSD is coupled to the sperm protein in a way that we have just never seen before, or even imagined. Its exciting to see how nature has done this and perhaps, in the future, we can learn from this to make synthetic proteins that can be turned-on by voltage or develop novel male contraceptives that work by blocking this protein.”
Source:
Journal reference:
Yeo, H., et al. (2023) Structure and electromechanical coupling of a voltage-gated Na+/H+ exchanger. Nature. doi.org/10.1038/s41586-023-06518-2.