Produced in Partnership with Park SystemsReviewed by Olivia FrostMar 25 2026
Understanding how living cells grow, respond to stimuli, and change over time is central to modern cell biology and drug discovery. Many traditional imaging approaches rely on fluorescent labels, which can introduce phototoxicity, bleaching, or unintended effects on cellular behavior. Digital Holographic Microscopy (DHM), a form of quantitative phase imaging, offers a powerful, label-free alternative that enables real-time monitoring of cellular morphology and intracellular dynamics over long time scales without disturbing the cell's natural state.
In this interview, Benjamin Rappaz from Lyncée Tec SA talks to NewsMed about how DHM complements fluorescence microscopy for live-cell imaging and high-content screening, and discusses practical considerations for assay design, data validation, and image analysis in quantitative phase imaging. The conversation also explores real-world applications in neuroscience and cardiotoxicity research, as well as the growing integration of DHM with other techniques, such as Atomic Force Microscopy, to support deeper insight into complex biological processes.
How do you define Digital Holographic Microscopy within quantitative phase imaging, and which label-free, non-invasive parameters does DHM quantify over long time scales in living cells?
Digital Holographic Microscopy (DHM) is an advanced quantitative phase imaging technology that enables real-time, label-free observation of living cells without altering their natural behavior.
Unlike fluorescence-based approaches, DHM does not require dyes or markers, allowing researchers to monitor biological processes over extended periods without phototoxicity or photobleaching. DHM quantifies key parameters such as cellular dry mass, optical volume, and refractive index, which are directly linked to cell growth, metabolism, and overall health status.
This makes DHM particularly valuable for long-term live-cell studies, where preserving the natural state of the cell is essential.
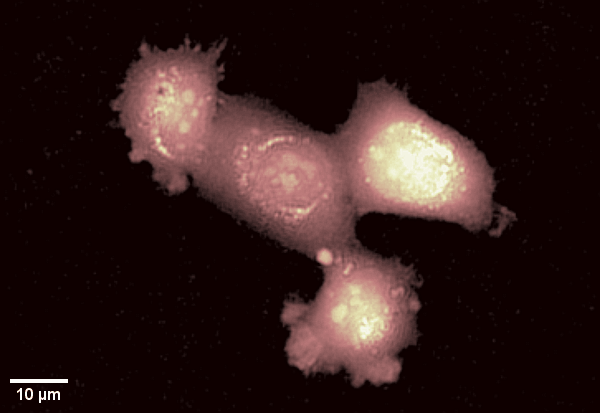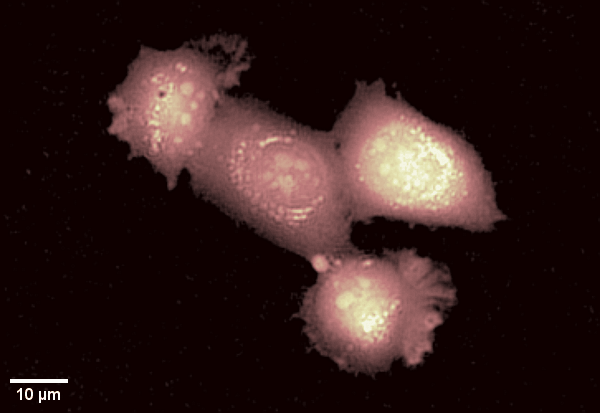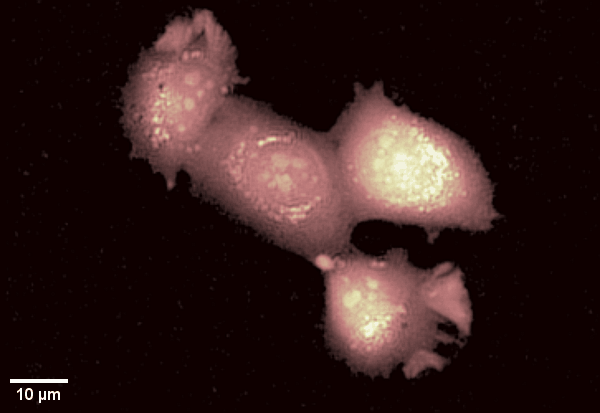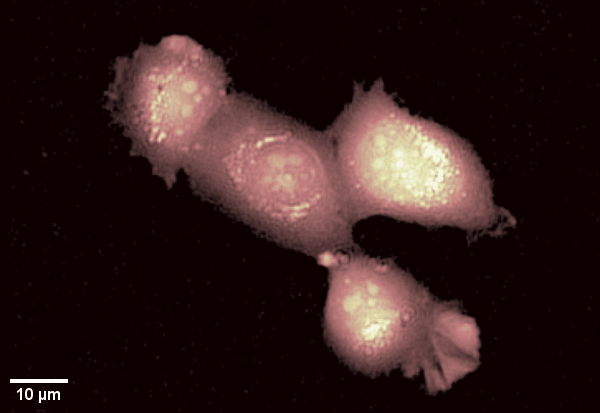
Human epithelial cell line imaged with Digital Holographic Microscopy. Label-free imaging allows for long time-lapse recording of dynamic membrane ruffling. Total time: 17 min. Image Credit: Park Systems
Where does DHM most clearly complement fluorescence microscopy in practice, and what information do you gain or preserve with DHM that fluorescence can obscure due to labeling, phototoxicity, or bleaching?
DHM and fluorescence microscopy are highly complementary techniques rather than competing ones.
Fluorescence provides molecular specificity, allowing researchers to identify and localize specific proteins or intracellular structures. In contrast, DHM offers continuous, quantitative insights into cell morphology and mass distribution without perturbation.
By combining both approaches, researchers can correlate dynamic morphological changes observed with DHM with molecular-level events captured through fluorescence. Importantly, DHM preserves cell integrity over long time scales, enabling observations that fluorescence alone cannot achieve due to phototoxicity or signal degradation.
This multimodal approach provides a more complete understanding of complex biological processes.
What validation frameworks are most effective for demonstrating concordance between DHM readouts, such as dry mass or refractive index, and orthogonal biochemical or fluorescence markers in assays intended for industrial or pharmaceutical partners?
DHM measurements such as dry mass and refractive index are based on well-established physical principles, ensuring high intrinsic accuracy.
For biological endpoints such as viability or drug response, validation is typically achieved through correlation with orthogonal methods, including fluorescence markers or biochemical assays. In practice, this involves establishing an initial calibration curve under controlled conditions, which can then be used to interpret DHM-derived metrics in a biologically meaningful way.
This combination of physics-based measurement and biological validation makes DHM particularly suitable for pharmaceutical and industrial applications, where reproducibility and data reliability are critical.
When scaling DHM for high-content screening, what have been the main bottlenecks for throughput and robustness, and how have you addressed plate-to-plate variability and drift over multi-day runs in collaborative projects?
DHM is particularly well-suited for high-content and high-throughput screening due to its fully automated workflow.
Processes such as plate scanning, autofocus, image reconstruction, cell segmentation, and quantitative analysis are integrated into a single pipeline, significantly reducing user intervention,variability and potential human bias.
Additionally, DHM is compatible with standard imaging plates, including plastic and glass-bottom formats, allowing easy integration into existing laboratory workflows.
This level of automation improves both throughput and data consistency, especially in multi-day experiments and collaborative research environments.
Can you walk through a concrete case study where DHM resolved a biological question in cell biology or neuroscience that fluorescence alone could not, and what was the decision-changing insight?
DHM has demonstrated unique value in applications where early, label-free detection is critical.
In neuroscience research, DHM enables the detection of early dry-mass redistribution events, which occur before visible morphological or fluorescence changes. This allows for earlier and more accurate prediction of long-term neuron viability.
In cardiotoxicity assays, DHM enables continuous, long-term monitoring of cardiac cell beating behavior. Subtle changes in rhythm or contractility can be detected at an early stage, providing valuable insight into drug-induced cardiotoxicity.
These examples highlight how DHM can reveal dynamic biological processes that are difficult to capture using fluorescence alone.
How do you approach multimodal correlative workflows, for example, pairing DHM with AFM, brightfield, or fluorescence, to link mechanical, morphological, and molecular phenotypes in the same cells?
Each imaging modality provides distinct advantages, and combining them enables a more comprehensive understanding of biological systems.
DHM offers fast, label-free imaging over a large field of view, capturing dynamic morphological and mass-related changes. In contrast, techniques such as AFM provide nanoscale resolution and mechanical insights.
By integrating DHM with AFM, fluorescence, or brightfield imaging, researchers can link morphological, mechanical, and molecular information within the same sample.
This multimodal approach is increasingly important for advanced life science research and drug discovery.
On the business and platform side, how has the integration of Lyncée Tec into Park Systems changed your product roadmap and the tangible outcomes customers see, such as throughput, ease of use, or new assays?
The integration of Lyncée Tec into Park Systems has significantly expanded both technological capabilities and global reach. With Park Systems’ established international sales and marketing network, DHM is now accessible to a broader customer base across academia and industry.
In addition, the integration enables closer alignment with AFM technologies, opening new opportunities for correlative workflows. For customers, this translates into improved usability, expanded application potential, and more comprehensive solutions for advanced research challenges.
Where are the strongest synergies between AFM and DHM for life science today, for example, instrument control, sample environments, or data fusion, and can you outline one end-to-end correlative workflow that a customer can deploy now?
DHM and AFM are highly complementary for life science because they operate at different but connected levels of observation.
DHM enables fast, label-free imaging over a large field of view, allowing researchers to monitor living cells dynamically and identify where meaningful biological changes are occurring over time. AFM, in contrast, provides nanoscale topographical and mechanical information with very high resolution.
The strongest synergy comes from using DHM for rapid, non-invasive screening and temporal monitoring, and then applying AFM to selected cells or regions of interest for deeper structural or mechanical characterization. Because both techniques can be applied in physiologically relevant sample environments, they are well suited for correlative life science workflows involving living cells.
A practical end-to-end workflow could begin with DHM to track cell growth, morphology, or dry-mass redistribution across a population over time. Once a specific cellular response is identified, AFM can then be used on the same sample to investigate membrane structure, surface topography, or nanomechanical changes at much higher resolution.
This kind of correlative workflow allows researchers to connect large-scale dynamic cellular behavior with localized nanoscale detail, providing a more complete picture of biological processes than either technique alone.
About Benjamin Rappaz 
Benjamin Rappaz is a life science researcher specializing in Digital Holographic Microscopy (DHM). He has been deeply involved in the development of high-content screening technologies, working closely with industrial and pharmaceutical partners to translate advanced imaging concepts into practical research tools. With a strong background in microscopy, image analysis, and cell biology, his work focuses on enabling researchers to study living cells quantitatively - without labels or complex sample preparation.
Expertise:
- High-content screening and assay development through academic–industry collaboration
- Advanced microscopy, including fluorescence and digital holography
- Cell biology, neuroscience, and molecular biology
- Project leadership and supervision of interdisciplinary research teams
About Park Systems
Park Systems is a global leader in nanoscale microscopy and metrology solutions, dedicated to empowering scientists, engineers, and industries to push the boundaries of innovation. Built on a pioneering legacy in atomic force microscopy (AFM), the company has grown into the world’s #1 AFM provider and now offers a diversified portfolio that includes imaging spectroscopic ellipsometry (ISE), nanoscale IR spectrometers (AFM-IR), white light interferometry (WLI), active vibration isolation (AVI), and digital holographic microscopy (DHM).
With headquarters in Korea, 16 global offices, and a trusted customer base of leading universities, national labs, and semiconductor fabs, Park Systems is shaping - and expanding - the future of nanometrology.