But stem cells can also be found in cancerous tissues, playing a role in the progression and metastasis of cancer.
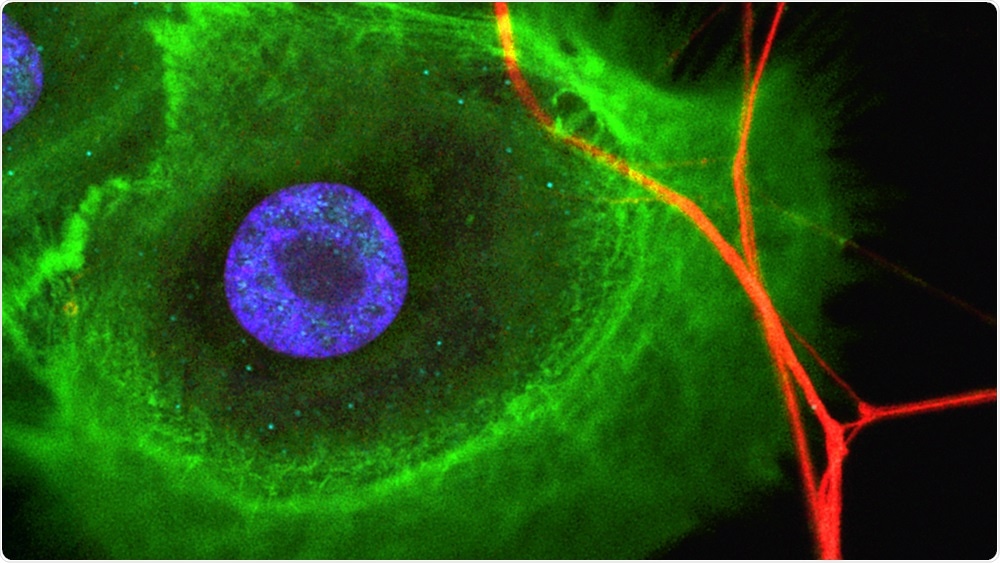
Cancer cells (green) are innervated with nerve cells (red). The cell nuclei (blue) are also visible. Image Credit: Institute of Oral Biology, UZH.
Nerves are integral for controlling the regenerative and physiological processes in which stem cells are also involved. But not much is known about the interactions that take place between the neurons and stem cells, specifically in cancers and in the regeneration of tissues.
Comparing stem cell types in tissue regeneration
Now, a research team, headed by Thimios Mitsiadis, professor at the Institute of Oral Biology of the University of Zurich, has reported a couple of studies that describe the way stem cells support the growth of neurons in cancer progression and tissue regeneration.
In the first study, the interaction of neurons was compared with two types of human stem cell populations, such as bone marrow stem cells and dental pulp stem cells.
Both stem cells are capable of differentiating into different types of cells, such as cartilage, bone, and fat cells.
Human bone marrow stem cells are the standard for bone regenerative techniques and these are isolated from skeletal bones. In addition, extracted teeth are the source of dental pulp stem cells, representing a potential alternative.
Dental stem cells are highly innervated
The research team used “organ-on-a-chip” technology, which depends on small three-dimensional devices simulating the basic functions of human organs and tissues, showing that both types of stem cells supported neuronal growth.
However, the dental pulp stem cells produced stronger outcomes than bone marrow stem cells: They triggered the formation of more elongated neurons, created dense neuronal networks, and set up close contacts with nerves.
Dental stem cells produce specific molecules that are fundamental for the growth and attraction of neurons. Therefore, stem cells are abundantly innervated.”
Thimios Mitsiadis, Professor, Institute of Oral Biology, University of Zurich
The development of such extended networks and the establishment of several contacts indicate that dental stem cells develop functional connections with nerves of the face. “Therefore, these cells could represent an attractive choice for the regeneration of functional, properly innervated facial tissues.”
Pierfrancesco Pagella, Study Co-Author and Junior Group Leader, University of Zurich
Cancer stem cells also recruit neurons
In the other study, the team investigated the interaction between nerves and cancer stem cells occurring in ameloblastoma—a severe tumor of the mouth. First, it was shown that ameloblastomas exhibit stem cell properties and are innervated by facial neurons.
Upon isolating ameloblastoma cells and placing them in the “organ-on-a-chip” devices, they retained not just their stem cell properties but also attracted nerves and created contact with them.
“It appears that nerves are fundamental for the survival and function of cancer stem cells,” explained Pagella. “These results create new possibilities for cancer treatment using drugs that modify the communication between neurons and cancer stem cells. We hope this opens unforeseen paths towards effective therapies against cancer,” added Mitsiadis.
The combination of advanced molecular and imaging tools and ‘organ-on-a-chip’ technology offers an exciting opportunity to reveal the hidden functions of neurons and their interactions with various stem cell types, in both healthy and pathological conditions.”
Thimios Mitsiadis, Professor, Institute of Oral Biology, University of Zurich
Source:
Journal reference:
Pagella, P., et al. (2020) Ameloblastomas Exhibit Stem Cell Potential, Possess Neurotrophic Properties, and Establish Connections with Trigeminal Neurons. doi.org/10.3390/cells9030644.