According to a new study by researchers from Kanazawa University and Tsukuba University, which was reported in Nanoscale, the physical properties of extracellular bacterial membrane vesicles are considerably varied.
The properties of a single type of bacterium and different types were found to be extremely heterogeneous.
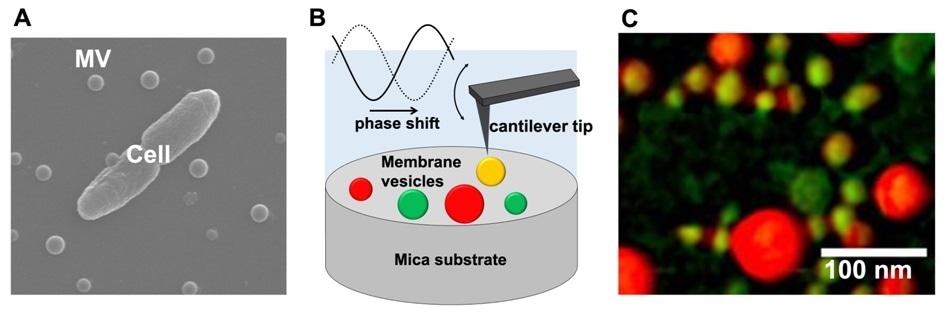
(A) Scanning electron microscopic image of a bacterial cell and extracellular membrane vesicles (MVs). (B) Schematic drawing of MVs observation using atomic force microscopy phase imaging. (C) Mapping of MVs’ physical properties using atomic force microscopy phase imaging. MVs are color-coded on a scale ranging from “non-adherent/hard” (reddish-colored spheres) to “adherent/soft” (greenish-colored spheres). Image Credit: Kanazawa University.
One feature of bacterial activity is the synthesis of extracellular membrane vesicles (MVs), which are biological packages enveloped by a lipid bilayer membrane and carrying genetic material. In addition to these specific biological functions, MVs have wider applications in the field of nanobiotechnology, such as drug delivery and enzyme transport.
For gaining better insights into the processes involving MVs, complete knowledge about their physical properties is crucial. Especially, the degree of heterogeneity of vesicles discharged by one type of bacterium is considered significant.
Azuma Taoka from Kanazawa University, Nobuhiko Nomura from Tsukuba University, and their collaborators have now demonstrated a hitherto unidentified physical heterogeneity in the membrane vesicles of four different types of bacterium.
The scientists employed phase imaging atomic force microscopy (AFM) to investigate the physical properties of MVs synthesized by Escherichia coli, Pseudomonas aeruginosa, Paracoccus denitrificans, and Bacillus subtilis.
Phase imaging AFM involves “tapping” a sample using a nano-sized oscillating cantilever tip. A comparison of the delay observed in the tip’s oscillation with free oscillation gives the measure of dissipation of energy upon its interaction with the sample surface.
This dissipation is, in turn, associated with the physical properties of the surface, such as elasticity, adhesion, and friction changes in which are caused by compositional differences.
Phase images of several MVs were recorded by Taoka, Nomura, and team, which were then color-coded on a scale ranging from “non-adherent or hard” (low elasticity, adhesion, and/or friction) to “adherent or soft” (high elasticity, adhesion, and/or friction).
The researchers found a higher diversity of physical properties of MVs by studying these maps. They checked for the changes in maps during imaging; the physical properties were found to be stable in time and it was concluded that diversity is an inherent feature of MVs.
The team identified that the physical heterogeneity was due to biological factors since shifts in the size and phase of MVs are not correlated. In addition, they found that different types of bacterium formed MVs with distinct physical-property distributions.
Finally, the researchers debated that the evident high heterogeneity shows that the chemical composition of MVs is heterogeneous.
The study by Taoka, Nomura, and colleagues not just offers a significant understanding of the properties of MVs produced by different bacteria, but also demonstrates the power of phase shift AFM as a tool for biological vesicles.
It is expected that using these cutting-edge techniques for nanoscale physical mapping will contribute to provide further detailed information to undiscovered nature of bacterial MVs and elucidate molecular mechanisms supporting their functions.”
Researchers of the Study
Source:
Journal reference:
Kikuchi, Y., et al. (2020) Diversity of physical properties of bacterial extracellular membrane vesicles revealed through atomic force microscopy phase imaging. Nanoscale. doi.org/10.1039/C9NR10850E.