Most often, cells are subjected to stressful conditions that can pose a serious threat to life, for example, toxins or high temperatures.
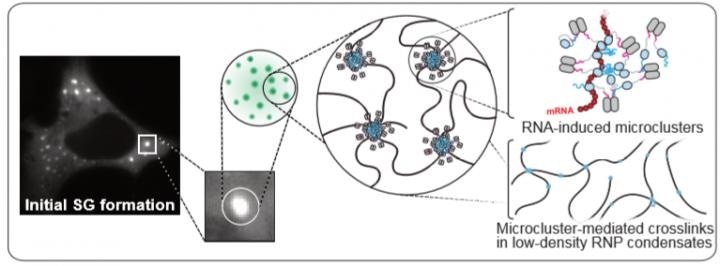
Model representing the behavior of the protein G3BP under stress: RNA molecules bind to G3BP, allowing multiple interactions that promote the clustering of G3BP and the assembly of dynamic stress granules (SG). The image on the left shows a cell in which stress granules were labeled with a fluorescent dye. The schematic on the right gives insight into the molecular structure and organization of stress granules. Image Credit: (C) BIOTEC.
Luckily, human cells are masters in managing stress using a strong response program: they stop to grow, create stress-protective factors, and develop massive structures, which are known as stress granules.
Researchers from Technische Universität Dresden (TU Dresden)’s Biotechnology Center (BIOTEC) and the Max Planck Institute of Molecular Cell Biology and Genetics (MPI-CBG), along with associates in Heidelberg and St. Louis (United States), have analyzed how these enigmatic structures are able to assemble and dissolve, and what are the factors that potentially make them change into a pathological state, as seen in neurodegenerative diseases like amyotrophic lateral sclerosis (ALS).
The study results were published in Cell—a leading scientific journal.
So far, ALS has remained an incurable disease of the central nervous system, where the motor neurons—that is, nerve cells that play a role in muscle movement—die slowly. But it is not known whether stress granules are implicated in this process.
Stress granules develop in the cell’s cytoplasm and assemble from a huge number of macromolecular components like RNA-binding proteins and messenger RNAs.
Generally, stress granules disassemble when there is a reduction in stress, a process that is supported by the powerful dynamic nature of stress granules. But the presence of persistent, non-dynamic forms of stress granules is a hallmark of ALS.
In ALS, patients suffer from muscle weakness and paralysis. Stress granule-containing motor neurons slowly degenerate, causing a progressive loss of motor functions. We need to better understand the complex biology of stress granules in order to design and develop future therapeutic strategies that counteract the course of the disease. But the complex environment of the cells within an organism makes this difficult.”
Dr Titus Franzmann, Study Senior Author, Max Planck Institute of Molecular Cell Biology and Genetics
To thoroughly verify their hypotheses regarding the assembly of stress granules as well as the pathology that causes molecular variations, the researchers created a regulated setting employing an in vitro system with purified components that helped to reproduce the stress granules in a test tube.
The researchers noted the assembly of the stress granules in a step-wise manner and defined the crucial factors that are fundamental to their dynamics.
Stress granules have a very complex structure. Nevertheless, their formation depends primarily on the behavior of a single protein—the RNA-binding protein G3BP. This protein undergoes a critical structural change: Under non-stress conditions G3BP adopts a compact state that does not allow stress granules to assemble. But under stress, RNA molecules bind to G3BP allowing multiple interactions that promote the assembly of dynamic stress granules.”
Dr Jordina Guillén-Boixet, Study First Author, Technische Universität Dresden
Dr. Guillén-Boixet continued, “The subsequent transition from dynamic into a non-dynamic state, which may be caused for example by prolonged stress, may trigger the death of the motor neurons, as we can observe in the disease ALS.”
Started in 2015, the research project was headed by the Alberti research team at TU Dresden’s BIOTEC. The close collaboration of 23 researchers from the Washington University in St. Louis (United States), the European Molecular Biology Laboratory in Heidelberg, TU Dresden, and the Max Planck Institute of Molecular Cell Biology and Genetics in Dresden was important for the project success.
There is a number of remaining questions. Our experimental system at BIOTEC is now available for further testing and will be central to developing new diagnostics and therapeutics to combat neurodegenerative diseases such as ALS.”
Simon Alberti, Professor, Technische Universität Dresden
Source:
Journal reference:
Guillén-Boixet, J., et al. (2020) RNA-Induced Conformational Switching and Clustering of G3BP Drive Stress Granule Assembly by Condensation. doi.org/10.1016/j.cell.2020.03.049.