In spite of several years of research, scientists are still clueless about how herpesviruses penetrate the human cells.
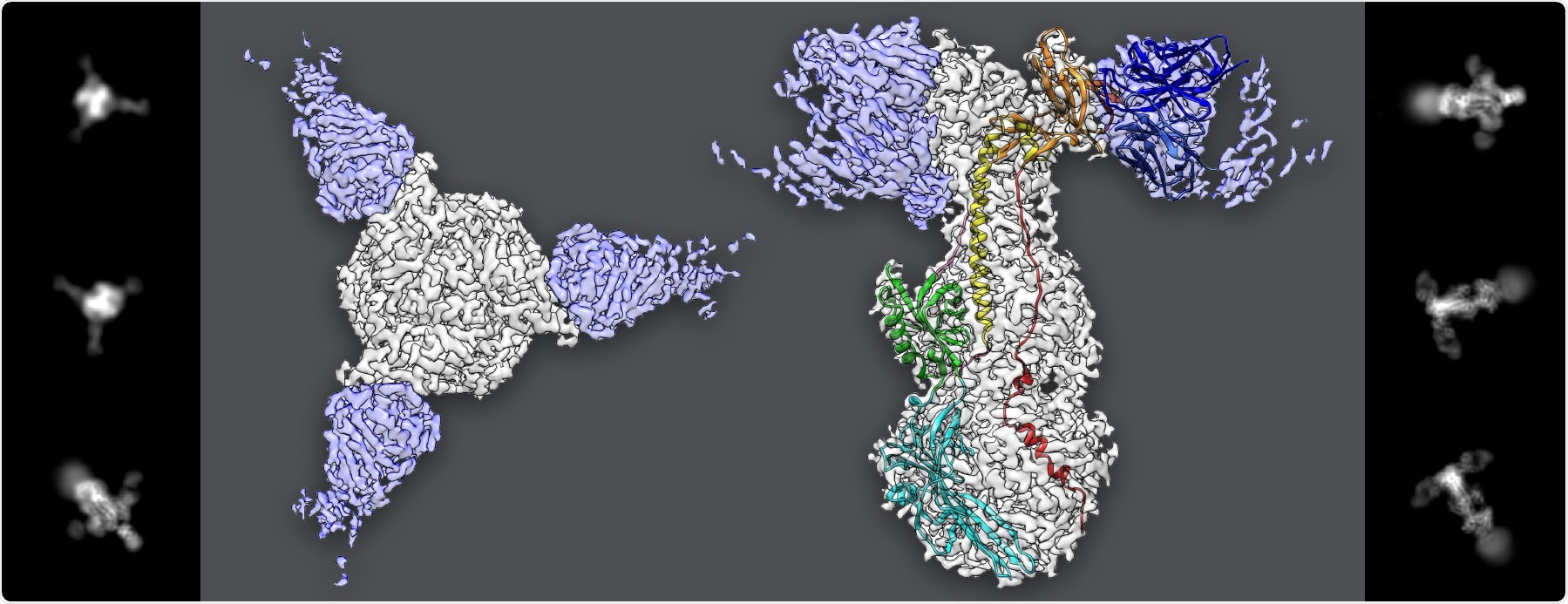
Images extracted from cryo-EM data (left and right panels) show the varicella zoster virus’ gB protein with three antibodies attached at the ends. Different views of the protein and antibodies together were used to reconstruct their molecular structure to nearly atomic resolution. Image Credit: Greg Stewart/SLAC National Accelerator Laboratory.
Now, scientists studying the varicella zoster virus (VZV), a type of herpesvirus that causes chickenpox, could have discovered a crucial clue: a major protein used by the virus to trigger infection does not work as previously believed, reported researchers from Stanford University and the SLAC National Accelerator Laboratory of Department of Energy.
The study was published in the Nature Communications journal on August 18th, 2020.
The study findings were achieved with the help of the high-resolution cryo-electron microscopy (cryo-EM) technique. The technique revealed that infections can be prevented by the immune system by attacking a site on the protein in an unforeseen location, stated Stefan Oliver, the first author of the study and a senior research scientist in pediatrics at Stanford University.
Herpesviruses, such as VZV—together with coronaviruses, HIV, and several other groups of viruses—are covered in a protective membrane.
To invade a specific cell, the viral envelope initially fuses with the membrane of the cell. In the case of the VZV virus, a protein known as gB that is present on the exterior of the viral envelope grabs onto and binds with the cells by using a set of molecular fingers.
As it turns out, that is only a part of the story. To find out what exactly is taking place in a more detailed way, Oliver and collaborators utilized an antibody isolated from a patient. In cryo-EM experiments, this antibody prevented the VZV virus from fusing with the cells to identify the site where the antibody attacks the gB protein.
To their amazement, the researchers observed that the antibody attached to a site on the gB protein was far from the fusion fingers, suggesting that it may not have to target the fusion fingers to inhibit fusion with a cell. This finding implies that more antibodies could be involved in the fusion process, which leads to infection, than was previously believed.
According to Oliver, understanding exactly how the process of fusion works will require more research works that could inform the design of vaccines and treatments for other types of herpesviruses because they also depend on the gB protein to infect cells.
Vaccines are currently not available for herpesviruses, with the exception of the one that prevents VZV, so the development of vaccines that target this newly identified region of gB has the potential to solve an important medical need.”
Stefan Oliver, Study First Author and Senior Research Scientist, Department of Pediatrics, Stanford University
“It was only possible to uncover this mechanism by generating one of the highest resolution structures of a viral protein-antibody pair using cryo-EM. Without the cryo-EM capabilities at SLAC these fascinating insights into the molecular mechanisms of fusion function would not have been achievable,” Oliver concluded.
Source:
Journal reference:
Oliver, S. L., et al. (2020) A glycoprotein B-neutralizing antibody structure at 2.8 Å uncovers a critical domain for herpesvirus fusion initiation. Nature Communications. doi.org/10.1038/s41467-020-17911-0.