The advancements in technology might come up with techniques that measure biological parameters in real-time in living organisms, which can help monitor tissue homeostasis, on the go. A significant component of such homeostasis is tissue oxygen concentration.
Scientists from Doshisha University have used two-photon phosphorescence lifetime imaging microscopy to measure oxygen concentration within osteoclasts of live mice. Through live oxygen measurements, they have further identified a novel response through oxygen-mediated DNA methylation to elicit osteoclastogenesis. Image Credit: Image courtesy: Keizo Nishikawa from Doshisha University.
The tissues have sufficient oxygen for carrying out biological functions in normoxic conditions. However, in hypoxic conditions—oxygen deprivation inducible in real life at the time of traveling in higher altitudes or by asphyxiation—the tissue might not have sufficient oxygen for such upkeep. This induces a hypoxic response. The cells concerned activate mechanisms that assure hypoxia mitigation.
To further analyze the complexities of cellular response to fluctuating oxygen concentrations in the living body, as an initial futuristic step, researchers from Doshisha University, Kyotanabe, successfully created a robust imaging technique comprising two-photon phosphorescence lifetime imaging microscopy.
They employed this microscopy technique in living mice to evaluate oxygen concentration in situ within multinucleated giant cells named osteoclasts that enable bone resorption. The observations were published in the journal EMBO Reports.
Through this study, we suggest a new approach to understand the physiological mechanisms of living organisms by accurately measuring the concentration of biomolecules deep in biological tissues. We believe ours is the first quantitative research attempt in the life sciences, similar to the that applied in the physical and chemical sciences to understand natural phenomena mathematically.”
Keizo Nishikawa, Study Lead Author and Professor, Graduate School of Life and Medical Sciences, Doshisha University
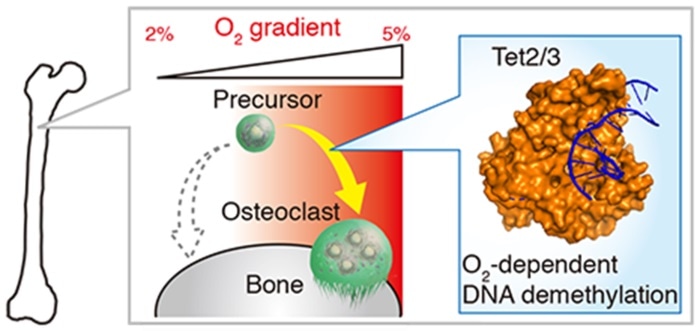
Through the current study, the researchers could evaluate the physiological range of oxygen tension in osteoclasts of live mice. The researchers noted that the range would be from 17.4 to 36.4 mmHg, in hypoxic and normoxic conditions, respectively. This indicates that physiological normoxia matches to 5% and hypoxia to 2% oxygen in osteoclasts.
Moreover, the independent examination of hypoxic conditions in bone tissue revealed that the shortage of oxygen gravely restricts osteoclastogenesis or the mechanism through which new osteoclasts grow. The researchers also discovered that hypoxia intervenes with the activity of ten-eleven translocation (TET) enzymes, which perform a major role in osteoclastogenesis through oxygen-dependent DNA methylation.
The researchers put forth that osteoclastogenesis might be limited independent of energy metabolism. The hypoxia-inducible factor activity occurs under conditions of 2% oxygen in osteoclasts.
In general, the research along with providing a novel imaging technique to evaluate oxygen concentration also identified a major molecular mechanism involved in osteoclastogenesis. The significance of TET enzymes in controlling osteoclastogenesis within the physiological range of oxygen tension might help reveal previously unreported processes of physiological response to oxygen perturbation.
We expect our findings to be applied in new medical technologies that predict disturbances of homeostasis and disease onset in the body.”
Keizo Nishikawa, Study Lead Author and Professor, Graduate School of Life and Medical Sciences, Doshisha University
The significance of sensing oxygen by cells and their adaptation to its readiness garnered recognition with the 2019 Nobel Prize in Physiology or Medicine. The recent study by researchers from Doshisha University assures to augment this increasing knowledge on what constitutes the quintessential cellular process keeping individuals alive.
Source:
Journal reference:
Nishikawa, K., et al. (2021) Osteoclasts adapt to physioxia perturbation through DNA demethylation. EMBO Reports. doi.org/10.15252/embr.202153035.