The IL-6 protein family has a poor reputation for promoting inflammation, arthritis, autoimmune disease, and even cancer. However, a new USC-led study published in Communications Biology reveals the significance of IL-6 and related genes in maintaining and regenerating cartilage in both joints and the growth plates that allow children’s skeletal growth.
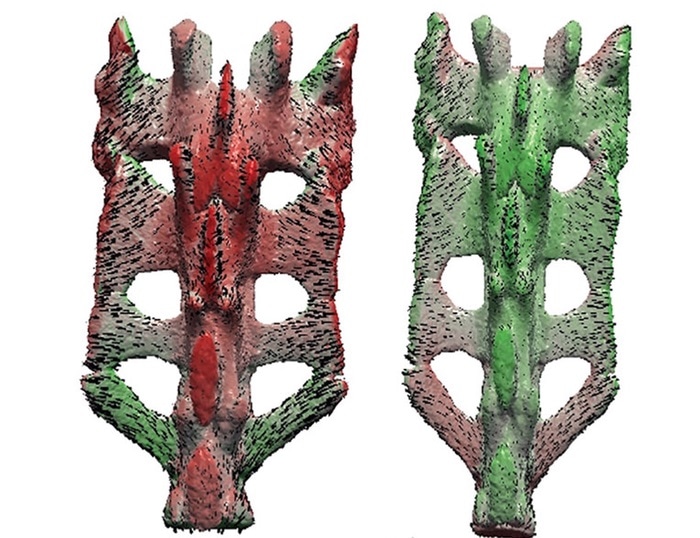
The spine from a healthy mouse (left) and a mouse with genetically disrupted cartilage progenitor cells. Image Credit: Dawei Geng and Tea Jashashvili.
We show, for the first time, that the IL-6 family, previously almost exclusively associated in the musculoskeletal field with arthritis, bone and muscle loss, and other chronic inflammatory diseases, is required for the maintenance of skeletal stem and progenitor cells, and for the healthy growth and function of the joints and spine.”
Denis Evseenko, Study Corresponding Author, Keck School of Medicine of USC
Denis Evseenko also adds, “Our study establishes a link between inflammation and regeneration, and may explain why stem and progenitors are exhausted in chronic inflammation.”
Evseenko is the J. Harold and Edna LaBriola Chair in Genetic Orthopedic Research, and an associate professor of orthopedic surgery, and stem cell biology and regenerative medicine at the Keck School of Medicine of USC.
The study’s first author, Nancy Q. Liu of USC, and her colleagues focused on STAT3, a key gene stimulated by IL-6. The researchers illustrated that STAT3 is required for the growth, survival, maturation, and regeneration of cartilage-forming cells in the joints and growth plates in both lab-grown human cells and mice.
When the gene stopped functioning, cartilage-forming cells became more dysfunctional over time, resulting in minimal body size, early fused growth plates, poorly developed skeletons, and moderately deteriorated joint cartilage.
Mice had the same problems when they failed to develop glycoprotein 130 (gp130), a protein that all IL-6 proteins use to stimulate STAT3. Disabling another gene, Lifr, which encodes a protein that collaborates with gp130 to recognize one of the IL-6 proteins known as Lif, resulted in similar but slighter skeletal and cartilage changes.
The researchers were able to recover normal growth plates in mice with a lack of gp130 by over-activating STAT3, but this also caused cartilage overgrowth, which resulted in other skeletal abnormalities.
Surprisingly, the scientists found significant gender differences: when STAT3 stopped working, females encountered more serious skeletal and cartilage changes than males. To figure out the reason, the researchers changed estrogen levels in mice and lab-grown pig cartilage cells. Estrogen increased the level and activity of STAT3, implying that females may increasingly depend on this gene.
The findings have clinical implications for the use of established drugs that inhibit STAT3 to reduce inflammation in autoimmune diseases, as these drugs may intrude with repair and maturation.
The Evseenko Lab, on the other hand, has used their knowledge of the nuances of STAT3 and genes involved and proteins to build a fully focused drug with the ability to regenerate joint cartilage without stimulating inflammation. Human clinical trials for this drug will begin soon.
Our findings really shift the paradigm and challenge the existing dogmas in the field about how IL-6, STAT3, and associated genes and proteins influence not only inflammation, but also regeneration.”
Denis Evseenko, Study Corresponding Author, Keck School of Medicine of USC
Source:
Journal reference:
Liu, N. Q., et al. (2022) gp130/STAT3 signaling is required for homeostatic proliferation and anabolism in postnatal growth plate and articular chondrocytes. Communications Biology. doi.org/10.1038/s42003-021-02944-y.