The telomeres, or ends of the chromosomes, increasingly shorten as people age. Recently, Salk researchers found that when telomeres are very short, they can still interact with the mitochondria, the cell’s powerhouses.
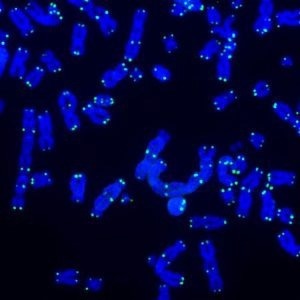 Telomeres protect the ends of chromosomes from damage. This image shows telomeres (green) and DNA (blue) during DNA repair activities. Click here for a high-resolution image. Image Credit: Salk Institute
Telomeres protect the ends of chromosomes from damage. This image shows telomeres (green) and DNA (blue) during DNA repair activities. Click here for a high-resolution image. Image Credit: Salk Institute
Initiating an inflammatory response that kills cells that could otherwise develop into cancer, this communication sets off a complicated network of signaling channels.
The research, which was published in Nature on February 8th, 2023, could help develop better therapies to counteract the negative effects of aging and innovative strategies for cancer prevention and treatment.
Co-senior authors and Salk Professors Jan Karlseder and Gerald Shadel worked together to examine commonalities they had uncovered in inflammatory signaling pathways. Their joint efforts led to the finding.
The telomere biology and how telomeres thwart the development of cancer are studied at Karlseder’s lab. The function of mitochondria in human disease, aging, and the immune system is studied at Shadel’s lab.
We were excited to discover that telomeres talk to mitochondria. They clearly synergize in well-controlled biological processes to initiate cellular pathways that kill cells that could cause cancer.”
Jan Karlseder, PhD, Study Co-Senior Author and Professor, Molecular and Cell Biology Laboratory, Salk Institute
Cells perish in a process known as “crisis” when telomeres shorten to the point that they can no longer shield chromosomes from harm.
This advantageous natural mechanism eliminates cells with unstable genomes and very short telomeres, and it is well-known to be an effective deterrent against the development of cancer. Karlseder and Joe Nassour, a senior research associate in the Karlseder lab and the study’s first author, previously found that the body rids itself of damaged cells using a process known as autophagy to eliminate cells in crisis.
The goal of this work was to determine how autophagy-dependent cell-death programs are triggered under times of stress when telomeres are exceedingly short.
The researchers identified interconnected immune sensing and inflammatory signaling pathways—similar to those used by the immune system to fight viruses—by performing a genetic screen on human skin cells known as fibroblasts which are essential for cell death during emergencies.
They specifically discovered that immune sensors termed ZBP1 and MAVS on the outer membrane of mitochondria are uniquely activated by RNA molecules coming from short telomeres.
The findings underline how cells can skip crisis (thus escaping elimination) and become malignant when the pathways are not working properly and show significant connections between telomeres, mitochondria, and inflammation.
Telomeres, mitochondria, and inflammation are three hallmarks of aging that are most often studied in isolation. Our findings showing that stressed telomeres send an RNA message to mitochondria to cause inflammation highlights the need to study interactions between these hallmarks to fully understand aging and perhaps intervene to increase health span in humans.”
Gerald Shadel, PhD, Study Co-Senior Author and Professor, Molecular and Cell Biology Laboratory, Salk Institute
Shadel is also the head of the San Diego Nathan Shock Center of Excellence in the Basic Biology of Aging and holds the Audrey Geisel Chair in Biomedical Science.
Joe Nassour, Study First Author and Senior Research Associate, Salk Institute, stated, “Cancer formation is not a simple process. It is a multistep process that requires many alterations and changes throughout the cell. A better understanding of the complex pathways linking telomeres and mitochondria may lead to the development of novel cancer therapeutics in the future.”
The researchers then intend to investigate the therapeutic possibilities of targeting these pathways to avert or treat cancer, in addition to the molecular basis of these pathways.
Source:
Journal reference:
Nassour, J., et al. (2023). Telomere-to-mitochondria signaling by ZBP1 mediates replicative crisis. Nature. doi.org/10.1038/s41586-023-05710-8