Scientists from The University of Texas at Austin, the University of California, Berkeley, and Korea University have improved one of the biggest scientific advancement of the past decade.
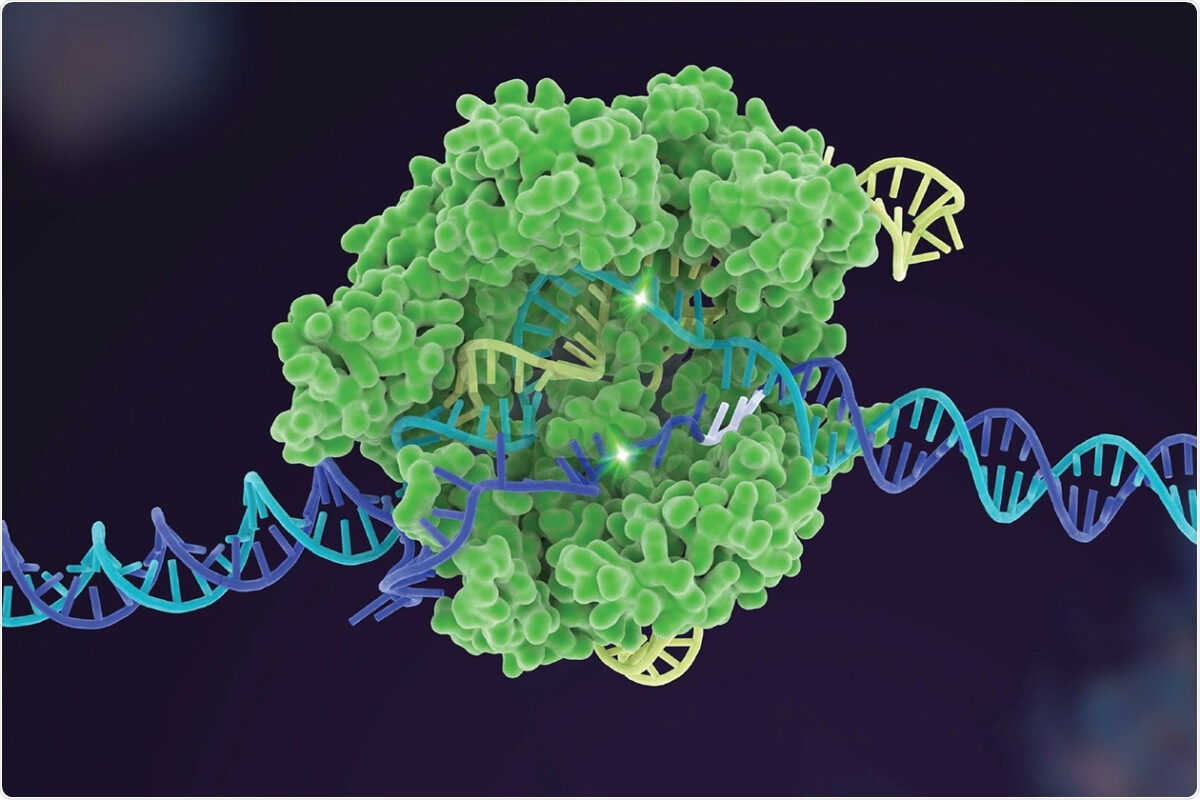
Image Credit: The University of Texas at Austin.
The researchers have designed a novel tool to help investigators select the best available gene-editing option for a specified task, rendering the CRISPR technology cheaper, safer, and more efficient. The new tool has been described in a study recently published in the Nature Biotechnology journal.
The CRISPR gene-editing method has enormous potential to enhance agriculture, human health, and the future of individuals on Earth; however, the fragile nature of gene editing poses a major challenge—there is virtually no room for errors.
Scientists edit genes by using scores of enzymes from a naturally occurring system, known as CRISPR. They identify a problematic DNA sequence and apply these specialized enzymes to snip, which is similar to using a pair of scissors. This method helps add, remove, or modify the genetic material.
However, these scissors are not ideal. Effectiveness and precision differ by the project and also by the CRISPR enzyme. The novel tool guides users, so that they can select the most optimal CRISPR enzyme for their high-stakes gene edit.
We designed a new method that tests the specificity of these different CRISPR enzymes—how precise they are—robustly against any changes to the DNA sequence that could misdirect them, and in a cleaner way than has ever been done before.”
Steve Jones, Research Scientist, The University of Texas at Austin
Jones co-wrote the article with Ilya Finkelstein, who is an associate professor of molecular biosciences.
When a CRISPR enzyme targets the wrong segments of DNA, it can lead to problems. Every CRISPR enzyme has strengths and weaknesses when it comes to editing varied sequences; hence, the team set out to develop a tool to help investigators make a comparison of different types of enzymes and identify the best one for a specified task.
CRISPR wasn’t designed in a lab. It wasn’t made by humans for humans. It was made by bacteria to defend against viruses. There is incredible potential for its use in medicine, but the first rule of medicine is ‘do no harm’. Our work is trying to make CRISPR safer.”
John Hawkins, PhD, Oden Institute for Computational Engineering and Sciences, The University of Texas at Austin
Hawkins is also an alumnus and was recently with the Oden Institute for Computational Engineering and Sciences at The University of Texas at Austin.
The researchers created a library of DNA sequences and quantified the accuracy level of every CRISPR enzyme, the time taken by the enzyme to modify the sequences, and the way they accurately edited the sequence. For certain tasks, the oft-used enzyme, known as CRISPR-Cas9, worked best, while for others, different enzymes performed relatively better.
It’s like a standardized test. Every student gets the same test, and now you have a benchmark to compare them.”
John Hawkins, PhD, Oden Institute for Computational Engineering and Sciences, The University of Texas at Austin
The new tool enables investigators to select the most optimal enzyme for editing on the first attempt, and hence, the process turns out to be cheaper and more efficient. It also provides information to scientists about where mistakes could probably occur for every enzyme and thus saves time.
“This technique gives us a new way to reduce risk. It allows gene edits to be more predictable,” Jones concluded.
Source:
Journal reference:
Jones Jr, S. K., et al. (2020) Massively parallel kinetic profiling of natural and engineered CRISPR nucleases. Nature Biotechnology. doi.org/10.1038/s41587-020-0646-5.