Immune cells are special cells ensuring the most effective defense against various pathogens, including viruses. Scientists from the University of Basel have gained insights into this specialization of T cells.
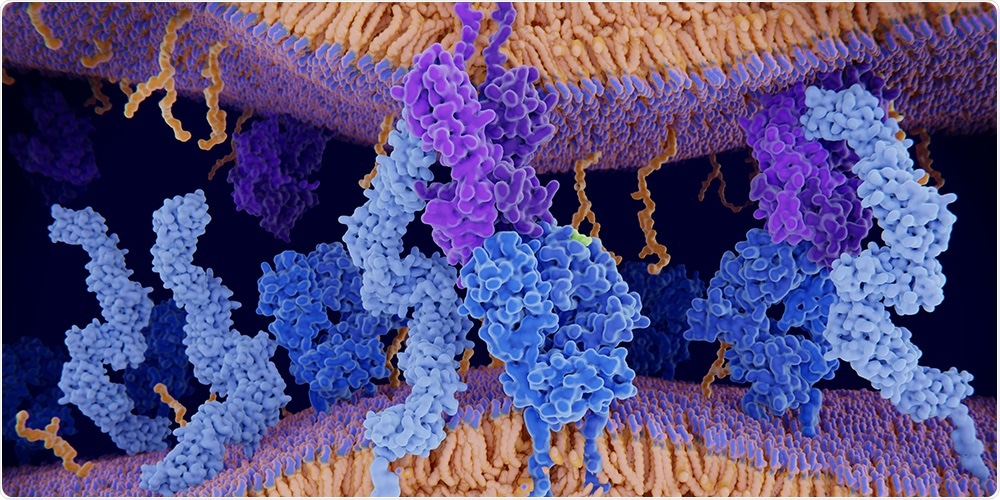
The T cell receptor (dark blue) is a kind of molecular sensor on the surface of the T cell that matches an antigen (piece of the pathogen, green) more or less precisely. The antigen is presented by a surface molecule (purple) of another immune cell called dendritic cell. Image Credit: iStock.
The researchers have demonstrated that it happens differently with regards to severe and chronic infection. This could inform new strategies to combat chronic viral infections.
Headed by Professor Carolyn King from the University of Basel, the researchers have devised a technique to analyze the specialization of T cells with respect to infections. A paper published in the eLife journal reports the various directions this specialization takes, based on whether it occurs in the context of an acute viral infection, such as influenza, or a chronic one, such as HIV infection or malaria, which can no longer be overpowered by the body.
The focus of the research was on the so-called T-helper cells. Activation of such a cell by a viral infection can allow it to specialize in one of two ways: into a Th1 cell that induces a more inflammation-heavy T killer cell response, or into a T follicular helper (Tfh) cell that mainly supports the production of antibodies. Thus, the balance between Tfh and Th1 cells has crucial implications for the inflammation level of the body’s immune response.
Degree of Activation Sets the Tone
There still exist gaps in the know-how of the way a T helper cell exactly decides which path to take during specialization. A factor that might have a crucial role is the strength of activation of the T helper cell—or, more accurately, its T cell receptor.
The T-cell receptor is a type of molecular sensor found on the surface of the cell that almost correlates with a part of the pathogen. The level of match is directly proportional to the level of activation of the receptor.
However, so far, the role of T-cell receptor signaling strength has been challenging to study in the context of viral infections. This is because researchers did not have an appropriate experimental model. King and her colleagues have now successfully developed one by joining hands with the research group of Professor Daniel Pinschewer, similar to King at the Department of Biomedicine.
Opposing Effects in Acute and Chronic Infection
The outcomes of the analysis astonished the researchers: T-cell receptor activation evidently has a role, but a contrasting one, based on whether the infection was chronic or acute. Strong activation resulted in more inflammatory Th1 cells in the case of acute viral infection. But in the case of chronic infection, strong activation synthesized more non-inflammatory Tfh cells.
The reason behind this could be an evolutionary adaptation of nature to protect its own body. If too many inflammatory Th1 cells are produced for too long during chronic infections, this would harm the body’s own organs in the long run.”
Dr Marco Künzli, Study First Author, University of Basel
Stronger for Longer
The experiments also offered another fascinating result: Th1 cells that formed due to weak activation could stay remain functional for a longer time in the case of chronic infection than if their specialization was activated by strong activation of the receptor. This is relevant since Th1 cells “fatigue” as time passes in the case of chronic infections.
Our results add a piece to the puzzle of T cell exhaustion and might—as a first step—contribute to new approaches in treating chronic viral infections.”
Carolyn King, Professor, University of Basel
Source:
Journal reference:
Künzli, M., et al. (2020) Opposing effects of T cell receptor signal strength on CD4 T cells responding to acute versus chronic viral infection. eLife. doi.org/10.7554/eLife.61869.