DNA methylation is a critical epigenetic alteration for mammalian development. DNA methylation, for example, is essential for numerous biological functions including gene control and cell destiny decisions. DNA methyltransferases help blastocysts re-establish global DNA methylation patterns upon implantation in animals.
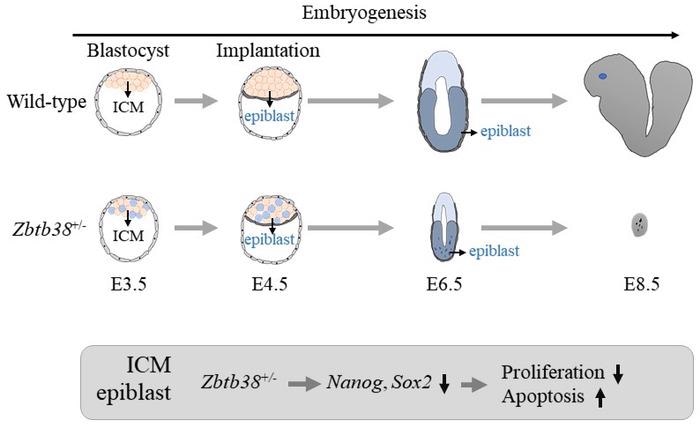 Heterozygous loss of the Zbtb38 (Zbtb38+/-) decreased epiblast cell proliferation and increased apoptosis shortly after implantation (E4.5~), leading to early embryonic lethality. Nanog and Sox2 expressions were reduced in the Zbtb38+/- embryos. ICM: inner cell mass. Image Credit: Eishou Matsuda
Heterozygous loss of the Zbtb38 (Zbtb38+/-) decreased epiblast cell proliferation and increased apoptosis shortly after implantation (E4.5~), leading to early embryonic lethality. Nanog and Sox2 expressions were reduced in the Zbtb38+/- embryos. ICM: inner cell mass. Image Credit: Eishou Matsuda
This is crucial for epigenetic data to be transferred to future generations. The involvement of methyl-CpG binding proteins (MBPs), which bind methylated CpG as part of the DNA methylation mechanisms, is, on the other hand, still unknown.
Zbtb38, also referred to as CIBZ, is a zinc finger type of MBP that is crucial for the proliferation of mouse embryonic stem (ES) cells, according to a prior study performed by researchers at the Nara Institute of Science and Technology (NAIST) in Japan. They also discovered that Zbtb38 promotes the expression of Nanog, which is essential for ES cell development. What Zbtb38 does in everyday life, though, remains a mystery.
The same researchers at NAIST, headed by Eishou Matsuda, employed Cre-loxP technology to create conditional Zbtb38 knockout mice in an attempt to answer the mystery. Their pioneering study found that a single Zbtb38 allele removal in the germline resulted in a reduction in epiblast cell development and an elevation in apoptosis shortly after implantation, resulting in early embryonic demise.
When Zbtb38 was deleted in heterozygous embryos, Nanog, Sox2, and other genes that regulate epiblast development and differentiation became abnormal.
Our findings indicate that germline loss of the Zbtb38 single allele reduces epiblast cell proliferation and increases apoptosis shortly after implantation, resulting in early embryonic lethality. Heterozygous Zbtb38 deficiency reduced the expression of Nanog, Sox2, and genes involved in epiblast proliferation, differentiation, and cell viability. This finding shows that a methyl-CpG binding protein has a role in controlling embryonic phenotype.”
Eishou Matsuda, Nara Institute of Science and Technology
“For the first time we demonstrated a link to an embryonic function for a protein that has long been known to bind methyl-CpG,” says study co-author Yasumasa Ishida.
This presents a huge opportunity for further research to find out how Zbtb38 works during embryogenesis. More research needs to be done to elucidate the specific molecular mechanisms. Zbtb38 is found in all tissues, and it is linked to height, cancers, neurodegenerative diseases and rheumatoid arthritis, etc.”
Eishou Matsuda, Nara Institute of Science and Technology
“Thus, the creation and analysis of tissue-specific Cre-mediated knockout mice will help us understand Zbtb38’s physiological functions and Zbtb38-linked diseases,” concludes Matsuda.
The findings of this study will be of interest to developmental biologists since they highlight the epigenetic importance of DNA methylation throughout the first trimester of pregnancy.
Source:
Journal reference:
Nishio, M., et al. (2022) Heterozygous loss of Zbtb38 leads to early embryonic lethality via the suppression of Nanog and Sox2 expression. Cell Proliferation. doi.org/10.1111/cpr.13215.