Most individuals are aware that DNA, or human genetic material, plays an important role in disease development, appearance, and susceptibility. This DNA is present in all cells and remains constant; when a cell splits to replenish itself, it creates an exact copy of its DNA. But if all cells begin with the same DNA, why do they not grow in the same way?
This is what scientists call an extremely outstanding topic, and in recent years, many scientists have focused on epigenetics, or how external stimuli can alter the expression of normally unchangeable genes within a cell for shorter or longer periods of time.
These factors include what humans consume, how they exercise, and how they age. Some experts believe that epigenetic modifications can be handed down from a parent to their unborn kid, thus programming the child to adapt to the environment in which the parent lives.
Some experts also contend that psychological factors in the parents, such as stress and trauma, might cause epigenetic alterations in an unborn child, such as heightened levels of stress hormones.
Epigenetic modifications are not irreversible and can be undone.
About the Project
The Novo Nordisk Foundation has awarded Jasmin Mecinovic 13,209,000 DKK for his project “Understanding the Chemical Language of Linker Histones.”
Molecules Form the Basis of All Life
All life is molecular at its core. So, the idea that organisms respond to influences at the molecular level is logical. I am a chemist, and I am interested in what happens at the molecular level when a specific epigenetic modification affects a gene.”
Jasmin Mecinovic, Professor, Department of Physics, Chemistry and Pharmacy, University of South Denmark
Epigenetic effects, whether natural or induced by environmental circumstances, cause chemical changes in particular structures surrounding DNA inside every cell nucleus. Histones are protein structures whose principal job is to create a framework around which the two-meter-long DNA strands can coil.
They do, however, have a metaphorical tail that goes beyond the DNA. Small chemical groups can be attached or removed from this tail by enzymes. When enzymes accomplish this, a signal is sent through the histone into the cell nucleus, indicating whether or not particular genes therein should be switched on or off.
A genome is an organism’s entire genetic material and includes a massive amount of information–most people have probably heard of the bases A, C, G, and T, which can be joined in an infinite number of ways between the two DNA strands.
Each link goes to a distinct gene, which governs the function of the cell in an organism. Simply put, the makeup of these bases is responsible for characteristics such as hair color, handedness, and the formation of additional toes.
Mecinovic added, “A human genome contains at least 20,000 genes, so let’s compare it to a book with 20,000 pages: It contains a lot of information, but not everything is in play at the same time. Let’s pretend that we both have a copy of that book. I open it at page 10, and you open it at page 100. We get two different pieces of information from the same source. That is how it works with genes; not everything is accessible all the time, only specific parts are activated. And this activation can be triggered by proteins that attach, remove or recognize small chemical tags on histones.”
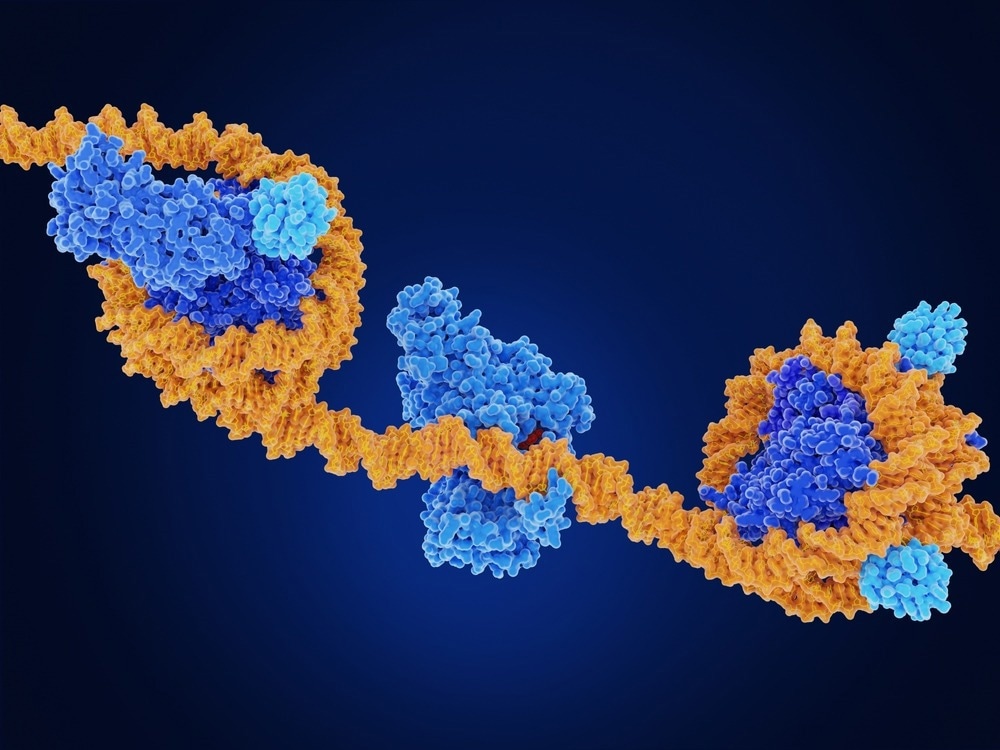
Image Credit: Juan Gaertner/Shutterstock.com
How and where enzymes attach chemical groups to histones and how such alterations influence gene expression is a tremendous riddle that researchers have only had 30 years to investigate. Since the alterations are so minute, incredibly sophisticated chemical instruments are required to peep into the realm of histones.
Fluttering Around with Spaghetti Arms
For the same reason, researchers have concentrated primarily on the four core histones that DNA strands coil around. However, there is a fifth histone termed linker H1, which is so chaotic that it was previously impossible to analyze using current approaches.
“Imagine that this histone has spaghetti arms that are constantly fluttering around, making it difficult to take a picture of it; it is like if I wave my hand while you take a picture, it becomes blurry. But this histone exists in humans and in other complex life forms, so it must serve an important function, and that is what I want to investigate,” Mecinovic further stated.
One of the primary goals of this new effort is to provide chemical methods for revealing what happens in the epigenetic corners of the erratic H1 histone.
Causing Diseases
H1, like the other histones, has a tail that extends beyond the DNA. Enzymes can also change it and, through the linker histone, affect genes in the cell nucleus.
Mecinovic concluded, “So, another core task will be to attempt to construct H1 histones that we can specifically modify to look like the natural H1, in order to see what effect it can have on interactions with histones, epigenetic proteins and DNA.”
A basic chemical study focuses on how H1 histone interacts with other biomolecules at the molecular level. Histones, on the other hand, can trigger unwanted activations or suppressions of genes, leading to disorders such as certain malignancies and autoimmune diseases such as rheumatoid arthritis, lupus, type 1 diabetes, and multiple sclerosis.