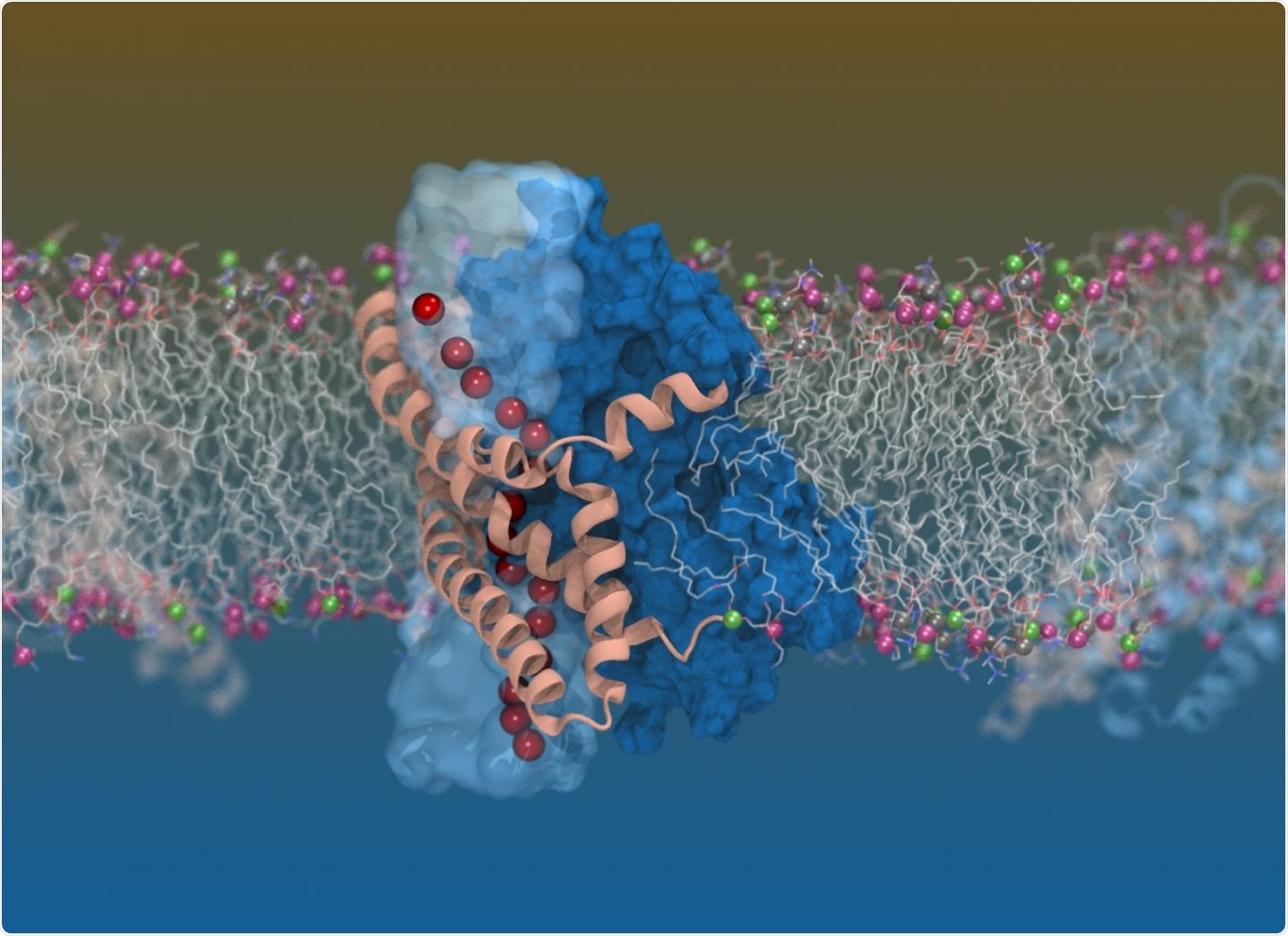
Computer simulation image of the glutamate transporter in our cells, created using images from cryogenic electron microscopy. Image Credit: Shashank Pant.
Glutamate transporters are essentially small proteins found on the surface of human cells that turn on and off the chemical signals. These signals play a huge role in maintaining the smooth function of cell-to-cell interactions. The glutamate transporters are also involved in nerve metabolism, signaling, and memory, and learning.
Using cryogenic electron microscopy (cryo-EM), the team captured the glutamate transporters in exquisite detail and demonstrated that they seem like a “twisted elevator” integrated into the cell membrane.
This first-ever discovery offers a whole new realm of possibilities, investigating if imperfections in the transporters could be the cause behind neurological disorders, including Alzheimer’s disease.
The study results have been published in the Nature journal.
The first time I saw the image was amazing. It revealed so much about how this transporter works and explained years of previous research.”
Ichia Chen, Study Lead Author and PhD Student, The University of Sydney
A multitasking transporter
The team was able to “photograph” the structure of the glutamate transporter, and they achieved this by studying an unlimited number of images confined in a thin ice layer using cryo-EM—an extremely sensitive microscope that enabled this study.
Using electron beams to image biological molecules, Cryo-EM can make things visible that otherwise cannot be seen with the naked eye. Moreover, the results confirmed the researchers’ long-held suspicions that glutamate transporters were multi-taskers.
Using Cryo-EM, we have uncovered for the first time just how these transporters can multitask - carrying out the dual functions of moving chemicals (like glutamate) across the cell membrane while also allowing water and chloride ions to move through at the same time.”
Renae Ryan, Study Senior Author and Professor, School of Medical Sciences, Faculty of Medicine and Health, The University of Sydney
Professor Ryan continued, “These molecular machines use a really cool twisting, elevator-like mechanism to move their cargo across the cell membrane. But they also have an additional function where they can allow water and chloride ions to move across the cell membrane.”
We have been studying these dual functions for quite some time, but we could never explain how the transporters did this until now. Using a combination of techniques including cryo-EM and computer simulations, we captured this rare state, where we can observe both functions happening at the same time.”
Renae Ryan, Study Senior Author and Professor, School of Medical Sciences, Faculty of Medicine and Health, The University of Sydney
Professor Ryan further added, “Understanding how the molecular machines in our cells work enable us to interpret defects in these machines in disease states and also gives us clues as to how we might target these machines with therapeutics.”
Key to bridging the gap in diseases
Comprehensive mapping out the structure of the glutamate transporter could be an important tool for scientists in interpreting the workings of the human body, and also the mechanism behind certain diseases.
Imperfections in the glutamate transporter have been linked to several neurological disorders, like stroke and Alzheimer’s disease. This comprises rare diseases, like episodic ataxia, a disease that impacts movement and leads to periodic paralysis, induced by an unregulated chloride leak via the glutamate transporter in brain cells.
“Understanding the glutamate transporter structure, which controls the normal flow of chloride, could help design drugs that can 'plug up' the chloride channel in episodic ataxia,” stated Dr. Qianyi Wu, the study co-lead author, from The University of Sydney.
Result of teamwork
The article was the outcome of seven years of research work from scientists in the United States and Australia. The study also emphasizes the potential and significance of high-resolution microscopy in interpreting biological processes.
“We are really excited to use the new Glacios Cryo-EM at the Sydney Microscopy and Microanalysis Facility, University of Sydney. Having access to this microscope 'in-house' will accelerate our research and our understanding of these important molecular machines,” Dr. Josep Font, the co-senior author of the study, concluded.
Source:
Journal reference:
Chen, I., et al. (2021) Glutamate transporters have a chloride channel with two hydrophobic gates. Nature. doi.org/10.1038/s41586-021-03240-9.